Mempro™ Detergent-Free FYVE Zinc Finger Protein Production
Based on the comprehensive protein engineering platform established through years of experience, scientists from Creative Biostructure offers tailored Mempro™ FYVE zinc finger production services using detergent-free expression system. FYVE zinc finger domain find in four proteins , Fab1, YOTB, Vac1 and EEA1 , which are four cysteine-rich proteins . FYVE domains combine Phosphatidylinositol 3-phosphate, and to some extent rely on its metal ion coordination and basic amino acids. By changing of pH, the FYVE domain plugs in cell membranes.
Endosome function and vacuolar protein sorting were related to the FYVE domain. The FYVE domain binds two zinc ions which are consisted of two small beta hairpins (or zinc knuckles) and an alpha helix in the behind. There are eight possible zinc harmonizing cysteine positions and are identified by having basic amino acids around the cysteines. Most of the members of FYVE zinc finger domain family also contain two histidines in a sequence motif. Nevertheless, the FYVE finger is structurally in common with the PHD finger and RING domain.
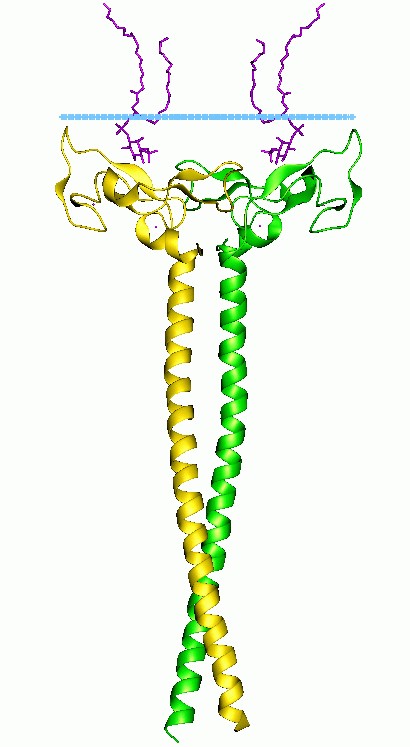 Figure 1. The structural of FYVE zinc finger domain (Mol.cell 2001)
Figure 1. The structural of FYVE zinc finger domain (Mol.cell 2001)
Creative Biostructure has rich professional experience in high quality FYVE zinc finger domain production using detergent-free membrane protein expression system, we can perform various strategies for Mempro™ detergent-free protein production, including:
- Mempro™ peptidase MA production using nanodiscs
Nanodiscs are composed of membrane scaffold proteins (MSPs), the gathered MSPs offer a phospholipid bilayer system. MSPs provide two surfaces, one is a hydrophilic at the outside, while the other is facing the lipids. Nanodiscs are modified by various of apolipoprotein A1 (apoA1), which is the primary ingredient in HDL. Moreover, the structure of Nanodiscs is extremely similar to high-density lipoproteins (HDL).
- Mempro™ peptidase MA production using amphipols
Amphipols (APols) are projected to maintain transmembrane proteins (TMPs) solvable in aqueous solutions without detergent, in the essence, it’s a family of amphipathic polymers.
- Mempro™ peptidase MA production using poly (styrene-co-maleic acid) lipid particles (SMALPs)
SMALPs are disc in the lipid bilayer polymerized by styrene maleic acid copolymer (SMA). These discs enable to assist the membrane proteins purified by using the tag of the protein in in their natural lipid surroundings.
These novel detergent-free technologies for FYVE zinc finger protein production can be obtained easily, and enabling more comprehensively structural and functional studies.
Creative Biostructure provides other various Mempro™ membrane protein production services. Please feel free to contact us for a detailed quote.
References:
C. Tribet, et al. (1996). Amphipols: Polymers that keep membrane proteins soluble in aqueous solutions. Proc Natl Acad. 93: 15047-15050.
M. Gaczynska, et al. (1994). Peptidase activities of proteasomes are differentially regulated by the major histocompatibility complex-encoded genes for LMP2 and LMP7. Proc. Nat. Acad. 91: 9213-9217.
M. Jamshad, et al. (2011). Surfactant-free purification of membrane proteins with intact native membrane environment. Biochem. 39: 813-818.
Lipoxygenases.
T. Bayburt, et al. (2002). Self-Assembly of Discoidal Phospholipid Bilayer Nanopar- ticles with Membrane Scaffold Proteins. NanoLetters 28: 853-861.