Recently, in a research report entitled “Structure of the human mediator bound transcription pre-initiation complex” published in the journal Science, scientists from Northwestern University and other institutions in the United States first observed multiple subunit machines responsible for regulating gene expression in human cells through research.
According to the researchers, the complex called Med-PIC (mediator-bound pre-initiation complex) is the key determinant of which genes are activated and which genes are inhibited. Mediators help to locate the rest of the complex (RNA polymerase II and general transcription factors) at the start of the genes that cells want to transcribe. Subsequently, the researchers used cryo-EM technology to visualize the complex with high resolution, which can help to understand the molecular mechanism of the complex’s function more deeply, because the complex plays a key role in the occurrence of many human diseases, Therefore, understanding its structural composition is crucial for developing effective therapies for these diseases, including cancer, neurodegenerative diseases, HIV, and metabolic diseases.
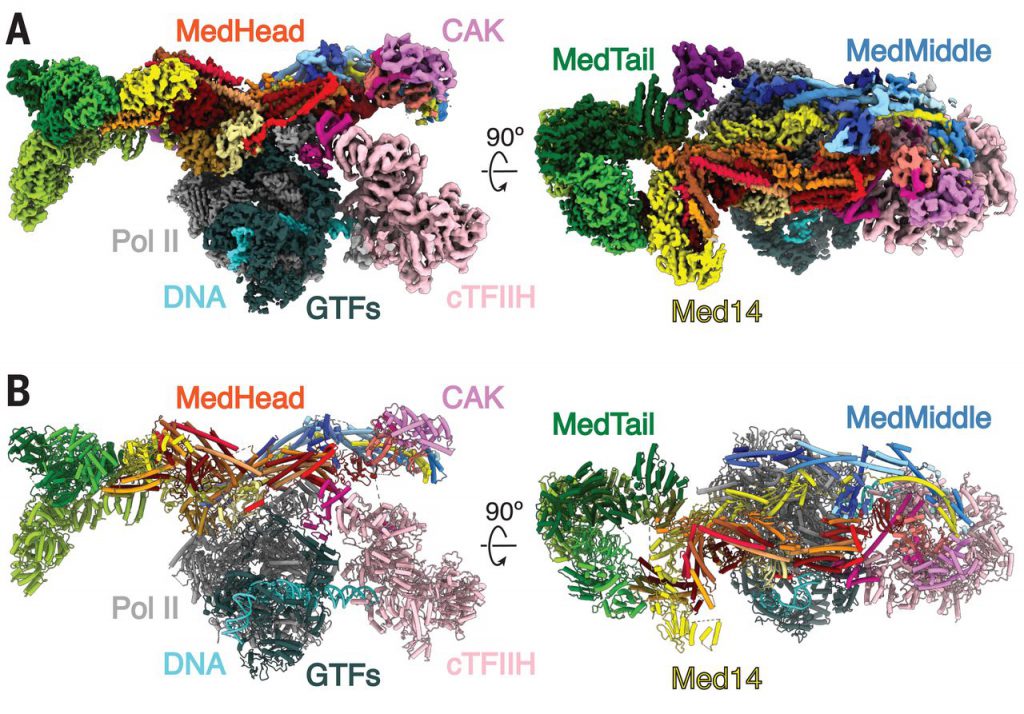
Researcher Yuan He said that in the context of gene expression, this “machine” is very basic for every branch of modern molecular biology. Visualizing its structure in 3D may help us answer basic biological questions, such as how DNA is transcribed into RNA. Observing its structure may help researchers understand its working mechanism. It’s like taking apart an ordinary household appliance to see how everything is put together. Now researchers can understand how the proteins in the complex are put together and function. In this study, the researchers observed the structure of the mediator complex in human cells for the first time in 3D.
The famous biochemist Roger Kornberg discovered the mediator complex in yeast in 1990, and then won the Nobel Prize in chemistry in 2006. However, the mediator contains 26 formidable subunits, and there are 56 of them combined with the promoter complex. Until now, researchers have only obtained the human version of high-resolution images. Roger Kornberg said that this is a very challenging research project in technology. These complexes are very rare, and it takes hundreds of liters of cells to obtain a small number of protein complexes.
In this study, researcher He and his colleagues found unexpected results when placing samples on a single layer of graphene oxide. Based on this support, graphene sheets can minimize the number of samples needed for imaging, and graphene may support a higher signal-to-noise ratio than amorphous carbon, which is typical support. When the samples are ready, the researchers use freeze electron microscopy to determine the 3D shape of proteins, which are usually thousands of times wider than the width of human hair. The principle of the technology is to shoot multiple two-dimensional images by emitting a beam of electrons on a rapidly frozen sample. In this study, researchers captured hundreds of thousands of images of the Med-PIC complex and then used computer methods to reconstruct its 3D images.
Researcher Talyzina explained that solving this complex problem is like assembling a jigsaw puzzle. Some of the subunits can be learned from other experiments, but we don’t know how the fragments are assembled or interact. With this final structure, we can finally observe the complete complex and understand how it is assembled. The resulting image shows that the Med-PIC complex is a flat, slender structure with a length of 45 nm. In addition, the researchers were surprised to find that the mediator moves relative to other parts of the complex, and it will bind to RNA polymerase II at a hinge point. Mediators are like pendulums. In the next step, researchers will continue to study and understand what this flexibility means. Researchers believe that this may have an important impact on the activity of key enzymes in the complex.
Reference
R. Abdella,A. Talyzina,S. Chen, et al. Structure of the human Mediator-bound transcription preinitiation complex, Science, 11 Mar 2021:eabg3074 doi:10.1126/science.abg3074