Understanding the principle of muscle movement can help us better fitness and weight loss. Understanding the movement principle of stem cells can help us better apply stem cell technologies in clinical practice.
For example, the hematopoietic stem cells of adult mammals reside in the “bone marrow”. This is why the transplantation of hematopoietic stem cells is usually referred to as “bone marrow transplantation”.
In actual bone marrow donation and transplantation, bone marrow transplantation does not change the bone marrow from one person to another, but uses the injection “mobilization needle” method to mobilize a large number of hematopoietic stem cells in the bone marrow into the peripheral blood to collect blood. That is, the movement of stem cells in the bone marrow microenvironment is involved.
Revealing how stem cells move in the human microenvironment can help us collect stem cells more scientifically and systematically, and understand how stem cells move. When whole blood is collected from a donor arm vein, hematopoietic stem cells can be efficiently extracted.
Similarly, when applying stem cells, understanding the movement laws of stem cells, intermittent movement, and intermittent contact with the microenvironment of damaged tissues, can provide a deeper understanding of stem cell mechanisms.
Development of in vivo imaging technology
In order to better observe the movement of stem cells, scientists from the Children’s Hospital of Boston and Massachusetts General Hospital in Boston, USA, jointly researched and developed live animal imaging technology of natural hematopoietic stem cells (HSC) and progenitor cells. This research result was published online in Nature.
Researchers created the mouse dual genetic technology, which refers to labeling the report label on a group of static long-term stem cells (LT-HSC) and is compatible with current live imaging methods in skull bone marrow.
Studies have shown that stem cells (LT-HSC) are close to the surface of the prevessel and the endosteum. In contrast, multipotent progenitor cells (MPP) vary more in distance from the intima and are more likely to be associated with transition zone vessels. Stem cells (LT-HSC) are not found in the most hypoxic bone marrow wall, but are found in a hypoxic environment similar to MPP.
In vivo, time-lapse imaging showed that the motility of stem cells in steady-state (LT-HSC) is limited. Activated stem cells (LT-HSC) are heterogeneous. Some cells become highly active, while some stem cells are clonally expanded in areas with limited space.
These structures have clear characteristics, because stem cell expansion is almost entirely found in the bone marrow cavity cell population with bone remodeling activity. In contrast, a cavity with low bone resorption activity will not accommodate the ever-expanding stem cells. These findings reveal the previously unknown fact that the bone renewal phase determines the heterogeneity in the bone marrow microenvironment. This method can directly visualize the behavior of stem cells and distinguish the heterogeneity of stem cells.
It is understood that biological research on hematopoietic stem cells is mainly carried out under transplantation conditions. According to scientists, studying dynamic stem cell behavior is challenging because it is not yet possible to visualize stem cells in living animals in their natural state.
Reveal the movement of stem cells in the bone marrow
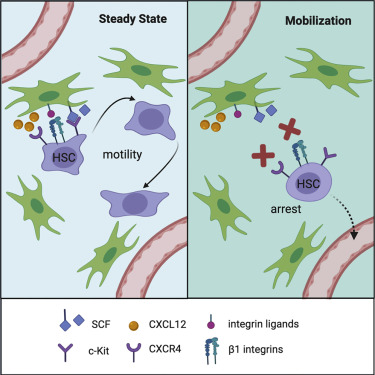
Researchers such as Boris Reizis of New York University and David R. Fooksman of Einstein College of Medicine have used in vivo imaging to reveal the motility of adult hematopoietic stem cells in the bone marrow microenvironment. This research result was published online on Stem Cell.
First, the researchers used a two-photon laser scanning microscope to observe genetically labeled hematopoietic stem cells in the bone marrow of mice and continued to observe for several hours. During this time, most stem cells exhibited a dynamic non-spherical morphology and significant motility. Hematopoietic stem cells move in the perivascular space and intermittently and closely contact the perivascular stromal cells.
In contrast, the blockade of CXCL12 receptor CXCR4 and integrin induced by mobilization quickly eliminated the motility and real-time shape dynamics of stem cells. These results reveal the dynamic changes of stem cell residence in the bone marrow and the interaction with the SCF+ matrix microenvironment, which will be interrupted when stem cells are mobilized.
At the same time, stem cells interact with BM microenvironment cells that produce growth factor c-Kit ligand (Kitl/SCF) and chemokine CXCL12.
Reference
Samik Upadhaya, Oleg Krichevsky, Ilseyar Akhmetzyanova, et al. Intravital Imaging Reveals Motility of Adult Hematopoietic Stem Cells in the Bone Marrow Niche. Cell Stem Cell, 27(2), 189-190, 6 August 2020, https://doi.org/10.1016/j.stem.2020.06.003