It has been known that most of the substances that are of significance to life are water-soluble, such as various ions and sugars. They need to enter the cells, and the water-soluble wastes produced during the life activities need leave the cells. Ion channels are made up of special proteins. They assemble and embed in the cell membrane. The pores form in the middle occupied by water molecules. These pores are channels through which water-soluble substances rapidly enter and leave cells.
The opening and closing of ion channels are called gating. According to the different gating mechanisms, the ion channels are divided into three categories: (1) Voltage-gated, also known as voltage-dependent or voltage-sensitive ion channels: opening and closing due to the changes of membrane potential. It is the easiest to pass ionic channel, such as K+, Na+, Ca2+, Cl-channel; (2) Ligand-gated, also known as chemically-gated ion channels: opening by the combining of transmitter and the binding site of the receptor, named by the transmitter receptors, such as acetylcholine receptor channel, glutamate receptor channel, aspartate receptor channel, etc.; (3) mechanical gating, also known as mechanical sensitivity ion channel: sensory of changes on cell membrane surface stress , to achieve extracellular mechanical signal transduction to intracellular.
The normal structure and function of cell ion channels is the basis for maintaining the life process, and its genetic variation and dysfunction are related to the occurrence and development of many diseases. We provide you with the latest research progress on ion channels to facilitate your research.
- Nature: TRP ion channel three brothers mediated acute thermal induction
doi:10.1038/nature26137
In a new study, researchers from Belgium’s Flemish Institute of Biotechnology (VIB) and the University of Leuven (KU Leuven) discovered three complementary ion channels in sensory neurons: TRPM3, TRPV1, and TRPA1, they mediate detection of harmful acute heat. The three redundant molecular thermal sensing mechanisms provide a powerful failure protection mechanism for preventing burns. The results of the relevant study were recently published in Nature.
These researchers first knocked out two different heat-activated TRP ion channels at the same time. It is known that one of them is also activated by capsaicin, an active ingredient in red pepper. But this only leads to a slight loss of heat-sensing. Interestingly, most of the remaining heat-sensitive neurons in these double knockout mice also responded to allyl isothiocyanate, which causes mustard, radish, and mustard to have a pungent taste.
This chemical selectively activated the third TRP ion channel, which prompted these researchers to further build triple knockout mice. These triple knockout mice completely lost the heat-induced pain response. Reintroduction of these three ion channels by transient transfection restored their thermosensitivity, and inhibitor cocktails against these three TRP ion channels also inhibited thermal reactions. This signaling is specific to thermally induced pain responses because although these mice usually respond to other pain stimuli (such as cold, stress, or acupuncture), their overall thermal preference is not affected.
- J Neurosci: Scientists discovered the reason for why if somebody is afraid of cold when he is young, he may have obesity when growing up!
doi:10.1523/JNEUROSCI.3002-17.2018
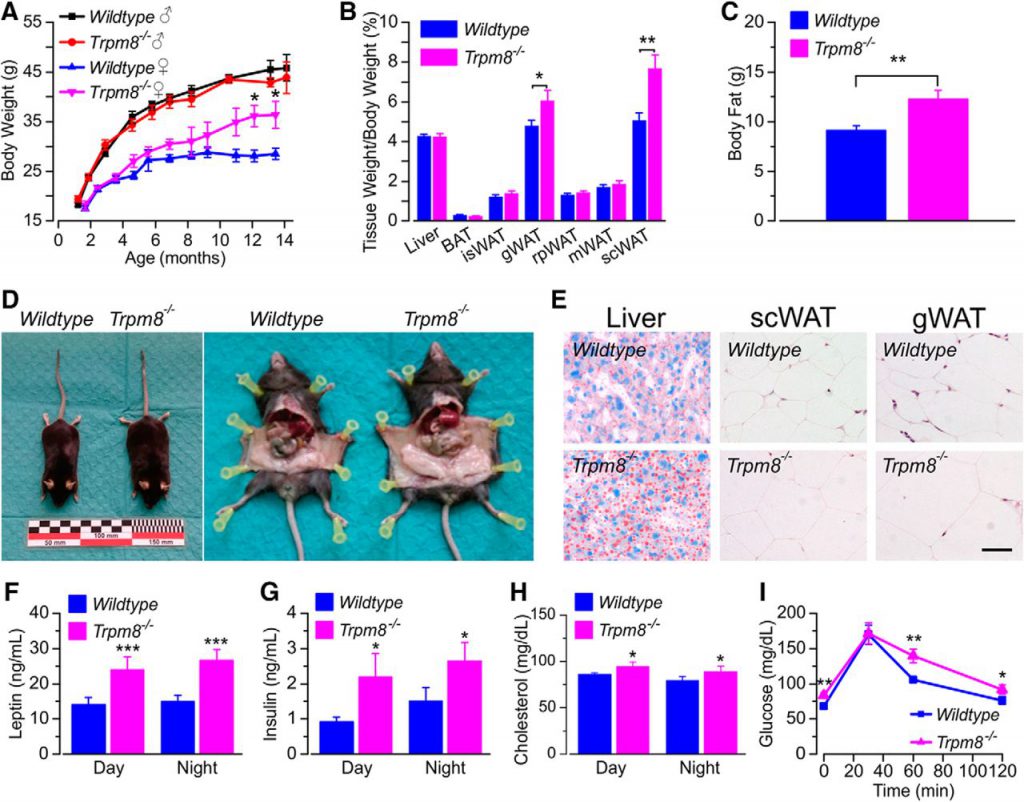
A recent study published in “J Neurosci” shows that the ability to control body temperature in cold environments may lead to fat growth in adulthood.
Rosa Señarís and colleagues from the University of Santiago de Compostela and the Institute of Neuroscience at Miguel Hernández de’ Elle University discovered that mice lacking the cold-sensing ion channel TRPM8, when they wake up during the day in a cold environment, they consume more food. This increase in daytime diet starts at a very young age, eventually leads to obesity in adulthood, and increases blood glucose, which may be due to reduced consumption of fatty. TRPM8-deficient mice lost more calories in the cold than mice in the control group, especially when fasting caused the body temperature to drop below 30°C.
This study explains the relationship between the cold and heat sensory system, heat regulation, and food intake and may lead to new ideas for the prevention and treatment of obesity.
- Nature: An in-depth understanding of the mechanism of acid-sensitive ion channels promises to develop new therapies for stroke and pain
doi:10.1038/nature25782
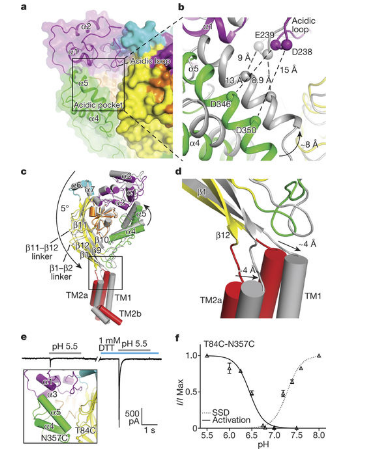
Recently, in a research report published in Nature, researchers from the Oregon Health and Science University (OHSU) revealed for the first time the atomic structure of key components of the nervous system.
In the article, the researchers used advanced imaging techniques to determine the resting state of an acid-sensitive ion channel. Dr. Eric Gouaux said that these ion channels are all over the body, which scientists often use as a key target of stroke therapy, it also plays an important role in pain transduction.
In this study, the researchers monitored the acid-sensitive ion channels independently. In the next phase of research, the researchers will study the channels embedded in the tissues to better understand how these ion channels interact with key proteins in the cell membrane. Nate Yoder said that to repair the car, we must understand the structure of the car engine. Based on the results of this study, later researchers will conduct more in-depth research to explore new therapies for the treatment of stroke and pain.
- Nat Genet: Scientists reveal the genetic mutations that cause rare hypertension syndrome!
doi:10.1038/s41588-018-0048-5
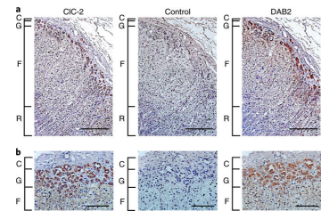
Twenty-five years ago, a rare genetic hypertension was discovered in a family in Australia. However, its genetic factors have not been clearly explained. By using modern sequencing methods, a research team headed by Ute Scholl, Professor of the Berlin Institute of Health, Johanna Kovant, successfully detected a new disease mutation (CLCN2) in the family and several other families. This mutation led to a familial hyperaldosteronism.
Ute Scholl and his team from the United States and Australia have now discovered a new gene for the pathogenesis of hypertension. The study focused on a special, very rare form of hypertension called type 2 familial aldosteronism. This genetic disease causes the adrenal gland to produce too much aldosterone, a hormone that regulates the body’s water and salt levels. Excessive hormones can cause high blood pressure.
Type II familial aldosteronism was first discovered and reported in Australia 25 years ago. The researchers investigated the family and other patients who were sick in children and even adolescents. Researchers analyzed a total of 8 families, including an Australian family, found a previously undetected genetic mutation associated with hypertension. The CLCN2 gene carries information that produces chloride channels on the cell membrane. In adrenal-specific cells, these chloride channels regulate membrane voltage and aldosterone production. These mutations in these patients caused changes in membrane potential and excessive production of aldosterone, which increased blood pressure.
- Science: Identifying a class of conserved gene family encoding proton-selective ion channels
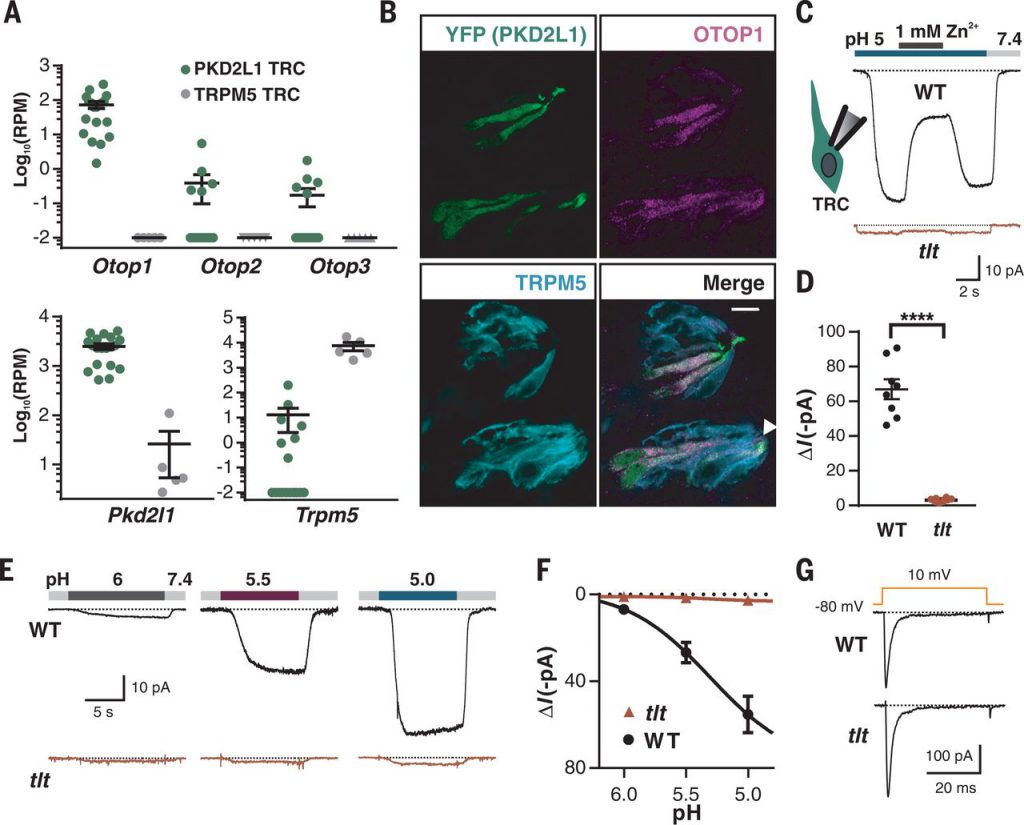
doi:10.1126/science.aao3264
In a new study, researchers from the Dornsife College of Letters, Arts and Sciences at the University of Southern California discovered a new class of ion channels. These channels allow protons (hydrogen ions) to enter the cell, play an important role in the balance of the inner ear, and are present in taste cells that respond to sour taste. The relevant research results were published in Science titled “An evolutionarily conserved gene family encodes proton-selective ion channels.”
Although genes encoding ion channels that allow protons to leave cells have been identified, it is not clear whether one or more genes are needed to form ion channels that allow protons to enter cells. Now, this research on sourness identifies the otopetrin gene family that encodes proton-conducting ion channels.
This gene family was originally thought to play an important role in maintaining body balance: Mutant mice, which change in the gene Otop1 (encoding otopetrin 1 protein) cannot keep themselves upright. The function of the protein encoded by this gene and the mutations that occur in it causing the defects of the inner ear vestibular system is not clear yet. But in the study of taste, a team led by Emily Liman, professor of biological sciences at the University of Southern California’s Donisf College of Arts and Letters, found that Otop1 encodes an otopetrin 1 protein that is a proton channel, and thus provide clues on how otopetrin 1 promotes inner ear function and maintaining balance.
In addition to Otop1, there are two related genes (Otop2 and Otop3) in vertebrates, and this gene family is also present in Drosophila melanogaster. There are structural differences between the otopetrin family and all other ion channels, and all otopetrin proteins form proton channels, suggesting that these proton-conducting channels are evolutionarily conserved. Each otopetrin protein is uniquely distributed in many tissues such as the tongue, ears, eyes, nerves, reproductive organs and digestive tract.
(To be continued…)
Reference
Vandewauw I, et al, A TRP channel trio mediates acute noxious heat sensing. Nature. 2018 Mar 29;555(7698):662-666. doi: 10.1038/nature26137
Reimúndez A. et al, Deletion of the Cold Thermoreceptor TRPM8 Increases Heat Loss and Food Intake Leading to Reduced Body Temperature and Obesity in Mice. J Neurosci. 2018 Apr 11;38(15):3643-3656. doi: 10.1523/JNEUROSCI.3002-17.2018.
Yoder N. et al, Gating mechanisms of acid-sensing ion channels. Nature. 2018 Mar 15;555(7696):397-401. doi: 10.1038/nature25782.
Scholl UI. et al, CLCN2 chloride channel mutations in familial hyperaldosteronism type II. Nat Genet. 2018 Mar;50(3):349-354. doi: 10.1038/s41588-018-0048-5.
Tu YH. et al, An evolutionarily conserved gene family encodes proton-selective ion channels. Science. 2018 Mar 2;359(6379):1047-1050. doi: 10.1126/science.aao3264.
Montell C, pHirst sour taste channels pHound? Science. 2018 Mar 2;359(6379):991-992. doi: 10.1126/science.aas9772.