Creative Biostructure has been in membrane protein production services for years. We have both scientific elites and ideal production platform to provide custom amyloid beta-sheet peptides using Mempro™ cell-based expression systems.
It is a representative Alzheimer’s disease histological feature that there is deposition of amyloid fibril on neuronal membranes. Amyloid beta-sheet peptides (Aβ) are peptides with 39-42 amino acids in length that are in close association with Alzheimer's disease. These peptides are cleaved productions of APP (amyloid precursor protein). Amyloid beta-sheet peptides can form flexible soluble oligomers with serval forms by self-aggregation. Some misfolded oligomers are thought to play an important role of inducing other amyloid beta-sheet peptides to adopt the misfolded oligomer formation as well, resulting in a chain reaction. Amyloid beta-sheet peptides are generally considered as intrinsically unstructured without a unique tetiary fold. Therefore we can not achieve structural knowledge on it by crystallization, making NMR and molecular dynamics as the major study area of the structure of amyloid beta-sheet peptides.
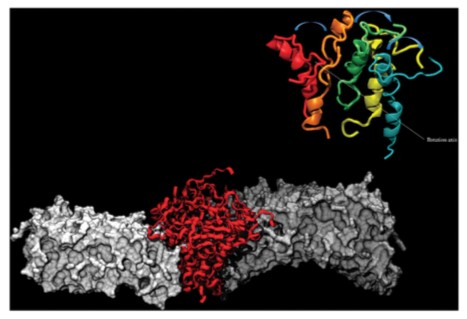
Figure 1. 24-mer Aβ aggregate inside a POPC membrane. (Phys. Chem. Chem. Phys., 2013)
Creative Biostructure can offer you the first-class membrane protein production services, and we can apply various strategies of Mempro™ cell-based protein production systems including:
• Mempro™ Amyloid Beta-sheet Peptides Production using Insect Cells System
Cultured insect cells and larvae are the most applied hosts for eukaryotic membrane protein production. Mempro™ protein production using insect cells system can sharply increase the expression in comparison with E. coli. Our advanced insect cells system can easily scale up and yield high-quality membrane proteins.
• Mempro™ Amyloid Beta-sheet Peptides Production using Mammalian Cells System
MemPro™ protein production in mammalian cells system facilitates to the correct folding and post-translational modification. We have proprietary MemPro™ protein production in mammalian cells system with high-quality eukaryotic membrane proteins from numerous mammalian cell types including HEK-293, COS-7, CHO and BHK-21.
• Mempro™ Amyloid Beta-sheet Peptides Production using Bacterial Cells System
Escherichia coli (E. coli) is the most widely applied bacterial host for membrane protein production. Lemo21 (DE3) strain is optimized for our Mempro™ protein production in our bacterial cells system. Other host systems using L. lactis, R. sphaeroides, and B. subtilis are also available for membrane protein production.
• Mempro™ Amyloid Beta-sheet Peptides Production using Yeast Cells System
Mempro™ membrane proteins production in Yeast expression system is one of the microbial systems. Our yeast cells system is an ideal host due because of following advantages: inexpensive media, fast growth rates, and high cell densities.
Creative Biostructure offers MemPro™ cell-based protein production services including expression, isolation, purification and crystallization of the membrane protein amyloid beta-sheet peptides. we are more than willing to help your structural and functional researches on amyloid beta-sheet peptides.
We offer various Mempro™ membrane protein production services. Please feel free to contact us for a detailed quote.
References:
E. G. Mufson, et al. (2016). Molecular and cellular pathophysiology of preclinical Alzheimer’s disease. Behav. Brain Res., 311: 54-69.
S. G. Itoh, et al. (2016). Oligomer Formation of Amyloid-β(29–42) from Its Monomers Using the Hamiltonian Replica-Permutation Molecular Dynamics Simulation. J. Phys. Chem. B, 120(27): 6555–6561.
K. L. Stewart, et al. (2016). Atomic Details of the Interactions of Glycosaminoglycans with Amyloid-β Fibrils. J. Am. Chem. Soc., 138 (27): 8328–8331
M. Pannuzzo, et al. (2013) Analytical model and multiscale simulations of Ab peptide aggregation in lipid membranes: towards a unifying description of conformational transitions, oligomerization and membrane damage. Phys. Chem. Chem. Phys., 15(23): 8940-8951.
