Protein Purification
The process that isolates one or a few proteins from cells or tissues is protein purification, which is vital for the characterization of the structure, function, and interaction of proteins. Protein purification is generally based on the distinction between protein size, properties, binding affinity, and biological activities. Creative Biostructure has developed an advanced purification platform that utilizes various purification strategies to provide either a small amount of protein for research purposes or manufacturing a large number of proteins for commercial uses. We are confident to efficiently deliver solutions for high purity protein purification and constantly develop and improve the purification protocols. The available methods to purify proteins range from simple precipitation to sophisticated chromatographic and affinity techniques.
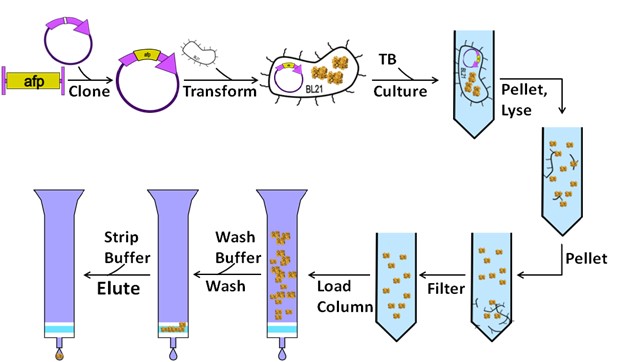 Figure 1. The workflow of protein
production and purification
Figure 1. The workflow of protein
production and purification
Depending on the project requirements and the nature of the proteins, purification protocols can be either de novo designed by us or obtained from customers or literature. Our purification methods include affinity chromatography with various tags, protein-specific chromatography, etc. The final goal is to accomplish a homogeneous and biologically active protein preparation that is critical for crystallization, protein-drug assays, and other related studies.
Protein Purification Protocols
- De novo purification protocols
- Existing protocols from customers or published sources
- Optimized protocols for various expression systems
Creative Biostructure's protein purification services include but are not limited to the following:
- Detergent Screening for Membrane Protein Purification
- Custom Affinity Chromatography Service
- Custom Ion-Exchange Chromatography Service
- Custom Size-Exclusion Chromatography Service
- Custom Hydrophobic Interaction Chromatography Service
- Custom Reversed-Phase Chromatography Service
- Custom TFF/Diafiltration Service
- Native Protein Purification
- Plasma Membrane Protein Isolation
Key Features
- Various target proteins: membrane-associated proteins, fusion and tagged proteins from recombinant sources, native proteins from natural source, enzymes, antibody/antigen
- Various sources: cultured cells, animal tissues, blood products, plant sources
- Protein purification scales from milligram to gram
- Protein refolding from inclusion bodies
- Proteins are purified according to client-specified purity
- Protein purification to GMP facility
With our expertise in protein chemistry, analytical chemistry, and functional assays, Creative Biostructure can deliver efficient and cost-effective solutions in protein purification; furthermore, we undertake the development of challenging proteins, such as co-expression or assembly of higher-order complexes. Creative Biostructure is also equipped with continuous fermenters and large-size chromatography systems allow the production of large amount of high purity proteins. After purification, protein analysis services including characterization, interaction, engineering, and structural determination are also offered by us. Please feel free to contact us for a detailed quote.
Ordering Process
References
- Gräslund S, et al. Protein production and purification. Nature Methods. 2008, 5(2): 135-146.
- Nehete J Y, et al. Natural proteins: Sources, isolation, characterization and applications. Pharmacognosy Reviews. 2013, 7(14): 107-116.

