Based on the comprehensive protein engineering platform established through years of experiences, scientists form Creative Biostructure offers unparalleled custom Mempro™ Bet v1-like protein production services using virus-like particles.
Virus-like particles (VLPs) can simulate the native virus, but are non-infectious since they do not have any viral genetic materials. VLPs have seclf-assembling multiprotein structures, which are commonly used in vaccine development. VLPs are commonly derived from the Hepatitis B virus (HBV). Recently, virus-like particles carrying conformationally-complex membrane proteins (termed as lipoparticles) have been applied for integral membrane protein production. Lipoparticles can incorporate a wide range of structurally intact membrane proteins, such as G-protein coupled receptors (GPCRs) and ion channels. Bet v1-like superfamily, which is a member of pathogenesis-related protein 10 (PR10), has a TATA binding protein (TBP)-like fold that containing beta-alpha-beta(4)-alpha structural domain. It is reported that Bet v1-like proteins are lipocalin-like proteins involved in lipid transport and/or metabolism.
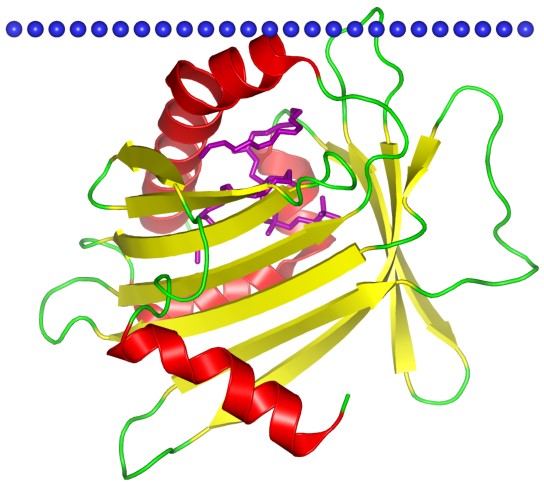 Figure 1. Phosphatidylcholine transfer protein, a Bet v1-like superfamily protein. (OPM database)
Figure 1. Phosphatidylcholine transfer protein, a Bet v1-like superfamily protein. (OPM database)
Virus-like particles can be applied for a large variety of applications, including:
- Antibody screening;
- Phage and yeast display;
- Immunogens/vaccines production;
- Ligand binding assays;
- Nucleic acids and small molecules delivery.
- Other potential applications
Creative Biostructure can provide Bet v1-like proteins in the stable, highly purified and native-conformation state. Lipoparticles can be produced from bacterial cells, yeast cells, insect cells, plant cells and mammalian cells for Bet v1-like protein production. Escherichia coli (E. coli) strains, insect cells and mammalian cells are the most widely employed for VLP production. For instance, we can obtain lipoparticles from mammalian cells by co-expressing the retroviral structural core polyprotein, Gag, along with a desired membrane protein. Gag core proteins self-assemble at the plasma membrane, where they bud off and capture membrane proteins of interest. Since the Bet v1-like proteins within lipoparticles are derived directly from the cell membrane without mechanical disruption or detergents, the native structure and orientation of Bet v1-like proteins are retained.
Creative Biostructure provides other various Mempro™ membrane protein production services. Please feel free to contact us for a detailed quote.
References:
A. Roldão, et al. (2010). Virus-like particles in vaccine development. Expert Rev. Vaccines, 9(10): 1149-1176.
F. Roth-Walter, et al. (2014). Bet v1 from birch pollen is a lipocalin-like protein acting as Allergen only when devoid of iron by promoting Th2 lymphocytes. J. Biol. Chem., 289: 17416-17421.
J. Chakrabortya and T. K. Duttaa (2011). From lipid transport to oxygenation of aromatic compounds: evolution within the bet v1-like superfamily. J. Biomol. Struc. Dynam., 29(1): 67-78.
S. Willis, et al. (2008). Virus-like particles as quantitative probes of membrane protein interactions. Biochemistry, 47(27): 6988-6890.