Protein Process Development and Manufacturing

Increasing demands on protein product quality lead to greater complexity of production process and analysis, therefore, require development and optimization of the production process. With the state-of-the-art technology and years of experiences in protein production, Creative Biostructure now provides custom process development and manufacturing services for recombinant soluble/membrane protein expression and purification. Our services emphasize on developing the optimal “from gene to protein” process for each individual case to meet your special requirements and objective in academic or industrial applications.
Upstream process development
- Protein and gene analysis. All information about the target protein can be helpful for the choosing of expression and purification techniques. We will take a comprehensive consideration of all the critical properties to help develop the optimal strategies, including but not limited to molecular size, isoelectric point, temperature stability, ionic strength stability, detergent requirement, biospecific affinity, etc.
- Protein recombination and expression. Specific affinity tag, vector, and expression system will be determined based on gene variety and requirements of future protein products. We provide histine-, MBP-, GST-, and other kinds of tagged recombinant protein vector construction, and various expression systems including bacteria, yeast, insect cells, plant cells, and mammalian for different applications.
We optimize growth and production media for target cell lines, determining feeding strategies, investigate cell line stability, and increasing production. We can perform specific process optimization studies depending on customer’s request.
Downstream process development
- Purification strategy and sample preparation development. Protein purification optimization includes increasing yield and improve product purity. Multiple advanced chromatography approaches and other techniques are available to choose at Creative Biostructure. Our protein science team will establish unique purification strategy for each individual case.
- Analytical assay development. We employ various analytical methods to check and maintain the purity of target proteins, including SDS-PAGE, mass-spectrometry, immune blotting, etc.
- Rapid technology transfer and scale-up to manufacturing. The protein platform at Creative Biostructure is equipped with GMP certified large-scale protein production facilities. Our manufacturing expertise, production capacity will help our customers to obtain competitive protein products in a cost-effective manner.
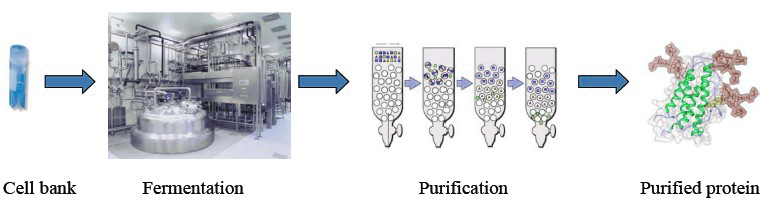 Figure 1. Protein manufacturing process
Figure 1. Protein manufacturing process
Creative Biostructure can scale up protein manufacturing from small quantities used for research to the much larger quantities needed for marketing. By outsourcing the process development of your projects to Creative Biostructure, you obtain access to a team of highly skilled and experienced staff, who will help to reduce your overall development risk. We ensure constant improvements in all aspects of biomanufacturing. Welcome to contact us for more details.
Ordering Process
Reference
- Gronemeyer P., Ditz R., Strube J., (2014). Trends in upstream and downstream process development for antibody manufacturing. Bioengineering. 1: 188-212.
- Meyer D., Chilkoti A., (1999). Purification of recombinant proteins by fusion with thermally responsive polypeptides. Nature Biotechnology. 17: 1112-1115.
