With state-of-art experimental systems and years of experience in the field of recombinant membrane protein production, Creative Biostructure provides advanced Mempro™ custom cell-based PGBD-like superfamily protein production service for various applications.
The PGBD-like superfamily contains member proteins or domains that have a common peptidoglycan binding function and a common core structure of a three-helical, closed bundle with a left-handed twist. According to the SCOP database, the PGBD-like superfamily is further catalogued into two families: the peptidoglycan binding domain (PGBD) family and the MMP N-terminal domain family. The PGBD family contains Zn2+ DD-carboxypeptidase (N-terminal domain) and N-acetylmuramoyl-L-alanine amidase YbjR (C-terminal domain). The MMP N-terminal domain family contains several members including Fibroblast collagenase (MMP-1), Gelatinase A (MMP-2), Stromelysin-1 (MMP-3) and Gelatinase B (MMP-9).
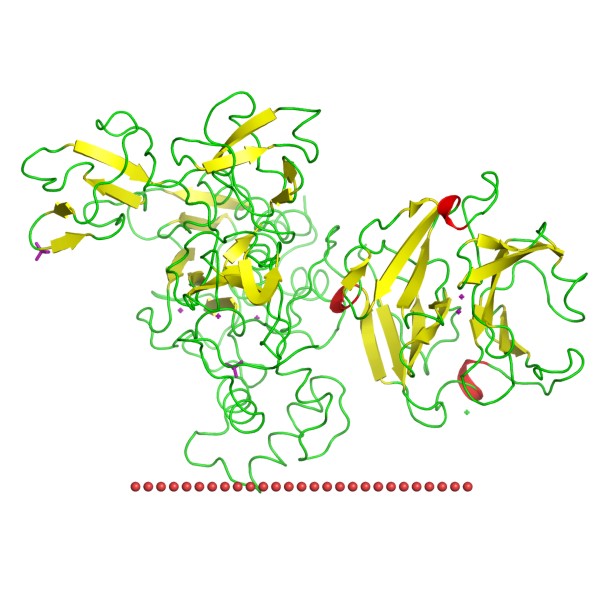 Figure 1. Structure of human 72 kDa type IV collagenase, full-length. (OPM)
Figure 1. Structure of human 72 kDa type IV collagenase, full-length. (OPM)
Creative Biostructure can provide the most advanced Mempro™ cell-based PGBD-like superfamily protein production service with high-quality and high-yield platforms. We can provide all kinds of cell-based expression systems to meet your special requirements, including:
Mempro™ Protein Production in Bacterial Cells System;
Mempro™ Protein Production in Yeast Cells System;
Mempro™ Protein Production in Incest Cells System;
Mempro™ Protein Production in Mammalian Cells System.
Cell-Based expression system is the most mature and widely applied platform for recombinant soluble/membrane protein production. Our science team will develop the optimal strategy according to the characteristics of target PGBD-like protein and your special requirements.
Bacteria systems (E. coli)
E. coli expression system dominates the bacteria systems and still remains to be the most popular expression systems in both research and industry applications. We will take the E. coli systems as our common first choice for its advantages of relatively low cost, fast, high yield, etc. Various bacteria expression vectors and E. coli host cells are available in Creative Biostructure.
Yeast systems
The yeast expression systems combine the advantages of being single cells, including easy genetic manipulation and fast growth, as well as eukaryotic features such as post-translational modifications (PTMs) and a secretory pathway leading to correct protein processing.
Insect cell systems
Insect cell expression systems are developed from transfected insect cells such as High Five, Sf9, and combined with vectors derived from baculovirus. Baculovirus systems are versatile and powerful tools for producing high levels of recombinant protein expression in insect cells. The baculovirus-insect cell expression systems have several advantages when compared with E. coli, including improved solubility, ability to incorporate PTMs, and higher yields for secreted proteins, etc.
Mammalian cell systems
The mammalian cell systems are the most powerful systems for mammalian or other eukaryotic proteins as they provide nearly-native intracellular environments for the expression of target proteins. They can produce a large amount of properly folded secreted proteins with the right glycosylation pattern. Despite the relatively high cost, cell lines derived from various mammalian cells such as CHO, BHK-21, HEK293, COS, HeLa and GH3 are still widely applied for integral membrane protein production.
Creative Biostructure can also provide a wide range of other custom Mempro™ membrane protein production services with multiple advanced technologies. Welcome to contact us for more details.
References:
Dideberg, O., Charlier, P., Dive, G., Joris, B., Frère, J.-M., & Ghuysen, J.-M. (1982). Structure of a Zn2+-containing D-alanyl-D-alanine-cleaving carboxypeptidase at 2.5 A resolution. Nature, 299, 469-470.
C. Trometer, and P. Falson (2010). Mammalian membrane protein expression in baculovirus-infected insect cells. Methods Mol. Biol., 601: 105-117.
S. Schlegel, et al. (2013). Bacterial-based membrane protein production. Biochim. Biophys. Acta., 1843(8): 1739-1749.