Creative Biostructure can offer high-quality custom Mempro™ phox homology (PX) domain-containing protein production services using cell-based expression system.
Mempro™ cell-based protein production systems are the most widely used systems to produce membrane proteins. PX domain is well known as a phosphoinositide-binding domain involved in targeting of proteins to cell membrane, which has a conseved motif of around 120 residues in length from human to yeast. PX domain is originally identified in p40phox and p47phox domains of neutrophilic NADPH oxidase complex. Proteins containing PX domain play important roles in cell signalling, lipid modification, protein sorting, and vesicular/membrane trafficking.
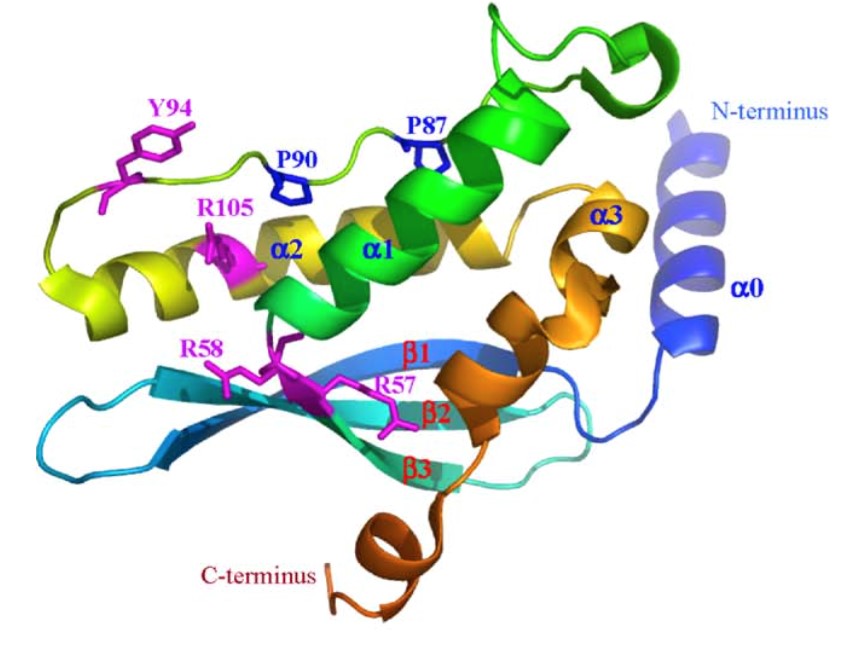 Figure 1. Illustrative structures of the PX domain of p40phox. (Biochim. Biophy. Acta, 2006)
Figure 1. Illustrative structures of the PX domain of p40phox. (Biochim. Biophy. Acta, 2006)
Creative Biostructure has extensive expertise in native conformation of PX domain-containg protein production using cell-based protein expression system, we can provide various strategies for Mempro™ cell-based protein production, including:
- Mempro™ Protein Production in Bacterial Cells System ;
- Mempro™ Protein Production in Yeast Cells System;
- Mempro™ Protein Production in Incest Cells System;
- Mempro™ Protein Production in Mammalian Cells System.
Among Mempro™ cell-based protein production platform, Escherichia coli is the most popularly used bacterial host for the production of PX domain-containing proteins. There are two general strategies can be performed in E. coli, the expression as soluble membrane proteins inserted into the membrane or as inclusion bodies representing cytoplasmic aggregates. Yeast cell system, combines prokaryotic as well as eukaryotic characteristics. It is a perfect eukaryotic host due to single cells, fast growth rates, inexpensive media, and high cell densities. Insect cells (such as Sf9, Sf21 and High Five) are also a major system for PX domain-like protein expression. Mammalian cells present another eukaryotic system for PX domain-containing protein expression, which provide nearly native-like cell environment. Cell lines derived from COS, CHO, BHK-21, HEK293, HeLa and GH3 are commonly used for integral membrane protein expression.
With the Mempro™ cell-based protein production platform, Creative Biostructure provides expressing, isolating, purifying and crystallizing PX domain-containing proteins to facilitate the study of their biological functions. Misfolding, aggregation, inactivity, poor stability and solubility are the common difficulties encountered in membrane protein production. Strikingly, we can overcome these difficulties by optimizing these factors like expression vectors, host strains, growth conditions, co-expression of helper proteins and fusion tags, etc.
We provide other various Mempro™ membrane protein production services. Please feel free to contact us for a detailed quote.
References:
C. S. Vollert and P. Uetz (2004). The phox homology (PX) domain protein interaction network in yeast. Mol. Cell Proteomics., 3(11):1053-1064.
C. Trometer, and P. Falson (2010). Mammalian membrane protein expression in baculovirus-infected insect cells. Methods Mol. Biol., 601: 105-117.
J. Petschnigg, et al. (2011). Using yeast as a model to study membrane proteins. Curr. Opin. Nephrol. Hypertens., 20(4): 425-432.
L. F. Seet and W. Hong (2006). The Phox (PX) domain proteins and membrane traffic. Biochim. Biophy. Acta, 1761(8): 878-896.
PX domain. (https://en.wikipedia.org/wiki/PX_domain)
S. Schlegel, et al. (2013). Bacterial-based membrane protein production. Biochim. Biophys. Acta., 1843(8): 1739-1749.
T. Lai, et al. (2013). Advances in Mammalian cell line development technologies for recombinant protein production. Pharmaceuticals, 6(5): 579-603.