With our scientific elites, Creative Biostructure has built an excellent and productive membrane protein production platform. Cell-based membrane protein production platform is the most used and advanced system to deliver our custom MemPro™ synuclein production service.
Synuclein family is a kind of common soluble proteins in vertebrates, mostly expressed in certain tumors and neural tissue. The synuclein family consists of three members known as alpha-synuclein, beta-synuclein, and gamma-synuclein. All three synucleins share a highly conserved lipid-binding motif within their sequences. The most famous member of synuclein family is alpha-synuclein. Alpha-synuclein has 140 amino acid residues, including three distinct regions. The membrane recognition region is located at the N-terminal, which is able to form an typical amphipathic helix conformation for membrane recognition and association. The abnormal aggregation of alpha-synuclein is closely related to Parkinson’s disease, and alpha-synuclein aggregates with amyloid β together as well. There are still several crucial questions about alpha-synuclein need to be answered including the structural basis for the conformational transition of alpha-synuclein.
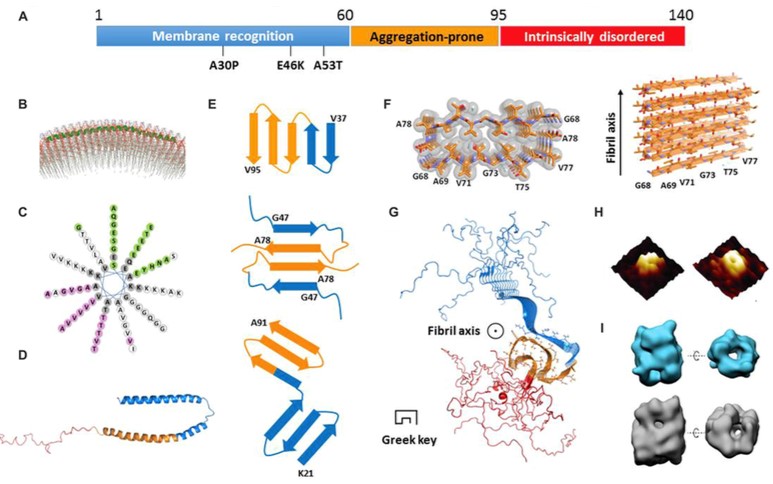
Figure 1. Different Structural models of alpha-synuclein. (Frontiers in Molecular Neuroscience, 2016)
Creative Biostructure can provide you the best membrane protein production services, and we can apply a lot of MemPro™ cell-based protein production systems including:
• MemPro™ Synuclein Production in Mammalian Cells System
Creative Biostructure can provide another great service-MemPro™ protein production in mammalian cells system. This system helps to correct folding and post-translational modifications for membrane protein. We have proprietary MemPro™ protein production in mammalian cells system with high quality eukaryotic membrane proteins from numerous mammalian cell types including human embryonic kidney cells (HEK-293), Chinese hamster ovary cells (CHO), monkey kidney fibroblast cells (COS-7) and baby hamster kidney cells (BHK-21).
• Mempro™ Synuclein Production using Insect Cells System
The most applied system for eukaryotic membrane protein production is in cultured insect cells and larvae. Mempro™ protein production using insect cells system can increase the expression level and reduce the truncated proteins compared to E. coli system according to the similar codon usage rules. Our innovational insect cells system based on baculovirus is advanced in several aspects as below:
• Proper folding and post translation modifications
• Easy scale-up
• Use of cell lines ideal for suspension culture
• High membrane protein yield
• MemPro™ Synuclein Production in Bacterial Cells System
Escherichia coli (E. coli) is the most widely applied bacterial host for membrane protein production. Lemo21 (DE3) strain is optimized for our MemPro™ protein production in our bacterial cells system.
• MemPro™ Synuclein Production in Yeast Cells System
Single-celled yeast is an easy, quick and economic culture host and able to apply multiple post translation modifications for eukaryotic membrane protein. Creative Biostructure has developed several strategies to improve yields per cell through optimizing the expression plasmid, host cell and culture conditions.
Creative Biostructure can offer MemPro™ cell-based protein production services including expression, isolation, purification and crystallization of the membrane protein synuclein. We are more than willing to help your structural and functional researches on synuclein.
We provide an array of MemPro™ membrane protein production services. Please feel free to contact us for a detailed quote.
References:
C. Wang, et al. (2016). Versatile Structures of α-Synuclein. Frontiers in Molecular Neuroscience, 9: A48.
C. D. Scala, et al. (2016). Common molecular mechanism of amyloid pore formation by Alzheimer’s β-amyloid peptide and α-synuclein. Scientific Reports, 6: A28271.
Z. Hu, et al., (2016). Membrane remodeling and mechanics: Experiments and simulations of alpha-Synuclein. Biochimica et Biophysica Acta-Biomembranes, 1858(7): 1594-1609.
