Creative Biostructure offers advanced custom Mempro™ protein production services for FYVE/PHD zinc finger proteins using cell-free expression system. Our patent cell-free expression systems can best preserve the native conformation of FYVE/PHD zinc finger proteins.
Zinc finger is a small protein motif that has a variety of diverse functions, including transcriptional activation, protein folding, DNA recognition, etc. zinc finger proteins always coordinate with one or more zinc ions to maintain their conformations. Based on the three dimensional structures, zinc finger protein could be classified into many different types.
Four cysteine rich proteins defined RYVE zinc finger: Fab 1 (yeast orthologue of PIKfyve), YOTB, Vac 1 (vesicle transport protein), and EEA1. FYVE zinc fingers are lipid binding proteins that involved in vacuolar protein sorting and endosome function. They recognize phosphatidylinositol 3-phosphate through a highly conserved motif. FYVE domain consists of two β-hairpins followed by an α-helix. The folding requires coordination with two zinc ions. The binding sites are defined by conserved cysteine residue and the basic amino acids close to cysteine.
The 3-dimensional structure of PHD fingers is very similar to RYVE fingers as they also contain a Cys4-His-Cys3 motif. The functions of PHD fingers are modification of chromatin and mediation of molecular interactions in gene transcription. PHD fingers not only recognize the lysine-methylated histone H3 but also the complex histone modifications. Since they control the gene expression by recruiting multi-protein complexes of chromatin regulators and transcryption factors, they are designated as epigenome readers.
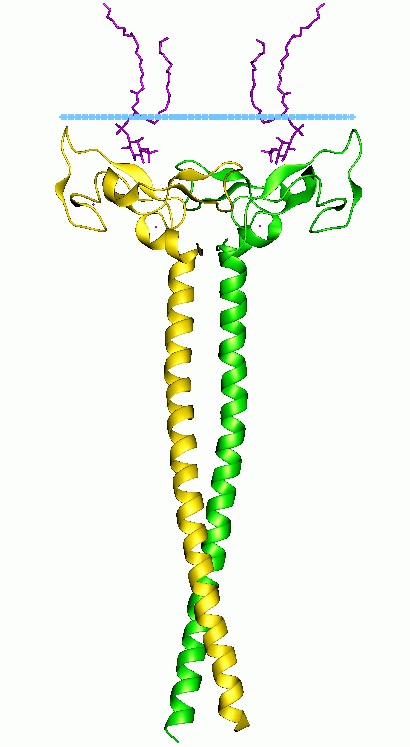
Figure 1. Early endosome antigen 1 (EEA1) dimer with lipids. (Wikipedia)
Creative Biostructure has a wealth of experience in high-yield FYVE/PHD zinc finger protein production using cell-free protein expression systems, we can provide various strategies for Mempro™ cell-free protein production, including:
- Mempro™ Cell-Free Protein Production in E. coli;
- Mempro™ Cell-Free Protein Production in Wheat Germ;
- Mempro™ Cell-Free Protein Production in Rabbit Reticulocyte;
- Mempro™ Cell-Free Protein Production in Baculovirus.
For high-yield FYVE/PHD zinc finger production, E. coli S30 extract system is the optimal system. It simplifies the process of transcription and translation from the DNA to protein. Mempro™ cell-free system developed by Creative Biostructure is deficient in protease which ensures the more stable protein production. FYVE/PHD zinc finger protein has been expressed in E. coli as a recombinant protein successfully. E. coli S30 extract system guarantees the FYVE/PHD zinc finger protein production with high purity and activity. In addition, Creative Biostructure also provides protein purification and crystallization service, which will help your project to move quickly and affordably.
We provide other various Mempro™ membrane protein production services. Please feel free to contact us for a detailed quote.
References:
F. Bernhard, et al. (2013). Cell-free expression - making a mark. Cur. Opin. Struct. Biol., 23: 374-380.
Laity J H, Lee B M, Wright P E. Zinc finger proteins: new insights into structural and functional diversity[J]. Current opinion in structural biology, 2001, 11(1): 39-46.