Creative Biostructure has proudly developed the unique protein engineering platform through years of experience, scientists from Creative Biostructure provide customized Mempro™ Acyl-CoA-binding protein (ACBP) production services using detergent-free expression system.
Acyl-CoA-binding protein (ACBP) is a 10 kD small protein, which is named as endozepine (EP) and diazepam binding inhibitor (DBI) as well. ACBP combines medium- and long-chain Acyl-CoA esters with very high affinity and it’s possible to function as an intracellular carrier of acyl-CoA esters. In addition, ACBP has the capacity to replace diazepam from the benzodiazepine (BZD) recognition site located on the GABA type A receptor. The protein probably serves as a neuropeptide to control the action of the GABA receptor. ACBP is extremely conserved and discovered in all four eukaryotic kingdoms, Fungi, Plantae, Protista, Animalia, and also found in several eubacterial species. Four alpha-helices form the ACB domain stand in a bowl shape with a exposed acyl-CoA-binding site. The residues on the protein are peculiar interact with the ligands and combine together.
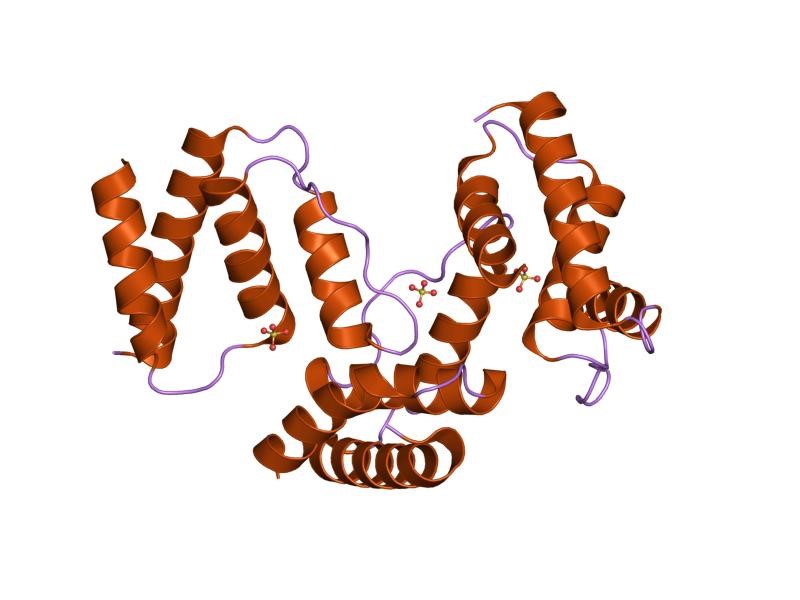 Figure 1. Structure of bovine Acyl-coA binding protein in tetragonal crystal form. (J. Mol. Biol. 2001)
Figure 1. Structure of bovine Acyl-coA binding protein in tetragonal crystal form. (J. Mol. Biol. 2001)
Creative Biostructure has ample professional experience in high quality ACBP production using detergent-free membrane protein expression system, we can carry out all kinds of strategies for Mempro™ detergent-free protein production, including:
- Mempro™ Acyl-CoA-Binding Protein (ACBP) Production Using Nanodiscs
Nanodiscs are soluble nanoscale phospholipid bilayers, which are made up of membrane scaffold proteins (MSPs). There are numerous membrane protein systems enable to be self-assembled into nanodiscs. In addition, the length of the MSP is the crucial factor of the size when the target membrane protein synchronously assembles with phospholipids into a discoidal bilayer.
- Mempro™ Acyl-CoA-Binding Protein (ACBP) Production Using Amphipols
Using amphipols for structural determination of membrane proteins is to maintain membrane proteins water-soluble by means of trapping them with amphipathic polymers instead of detergents. Amphipols (APols) meaning small amphipathic polymers which are enable to hold individual MPs water-soluble in their native state under the consist of little hydrophilic complexes.
- Mempro™ Acyl-CoA-Binding Protein (ACBP) Production Using Poly (styrene-co-maleic acid) Lipid Particles (SMALPs)
This approach generates SMALPs in which a little disk of lipid bilayer surrounded by polymer encircles the membrane proteins. It’s demonstrated that SMALPs may be a significant tool for the research of membrane protein structure and function.
These novel detergent-free technologies for ACBP production can be obtained easily, and enabling more comprehensively structural and functional studies.
Creative Biostructure provides other various Mempro™ membrane protein production services. Please feel free to contact us for a detailed quote.
References:
Bayburt, T. H., & Sligar, S. G. (2010). Membrane protein assembly into Nanodiscs. FEBS letters, 584(9), 1721-1727.
Jamshad, M., Lin, Y.-P., Knowles, T. J., Parslow, R. A., Harris, C., Wheatley, M., . . . Overduin, M. (2011). Surfactant-free purification of membrane proteins with intact native membrane environment. Biochemical Society Transactions, 39(3), 813-818.
Prabudiansyah, I., Kusters, I., Caforio, A., & Driessen, A. J. (2015). Characterization of the annular lipid shell of the Sec translocon. Biochimica et Biophysica Acta (BBA)-Biomembranes, 1848(10), 2050-2056.
Tribet, C., Audebert, R., & Popot, J.-L. (1996). Amphipols: polymers that keep membrane proteins soluble in aqueous solutions. Proceedings of the National Academy of Sciences, 93(26), 15047-15050.
