As an undisputed global leader in membrane protein production and manufacture, scientists from Creative Biostructure can offer customized Mempro™ Amyloid beta-sheet peptides production services using detergent-free expression system.
Amyloid beta-sheet peptides is a superfamily which consists of two families: Beta-sheet peptides of amyloid beta A4 protein and Beta-sheet peptides of major prion protein. Amyloid beta A4 protein, also known as APP or A4, belongs to Beta-sheet peptides of amyloid beta A4 protein family. This protein is composed of a large N-terminal extracellular region including heparin-binding and copper-binding sites, a short C-terminal intracellular domain and finally a short hydrophobic transmembrane domain. The N-terminal region of Amyloid beta A4 protein has an alike structure with cysteine-rich growth factors, while it probably serves as a cell surface receptor, which is conducive to neuronal adhesion, axonogenesis, neurite growth and cell mobility. Amyloid-beta40 (Abeta40) and amyloid-beta42 (Abeta42), two main isoforms of Amyloid beta A4 protein have been discovered in the extracellular brain deposits which are related to Alzheimer's disease (AD).
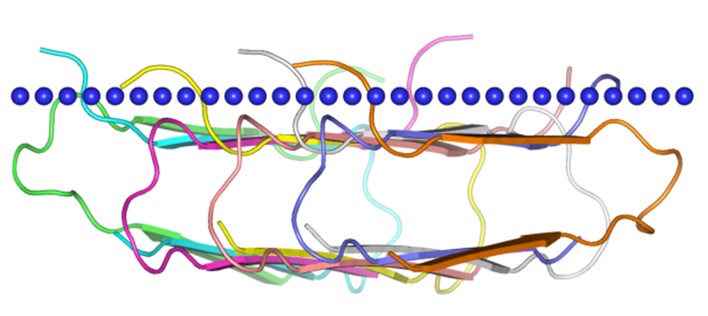 Figure 1. The structure of Amyloid beta A4 protein, peptide 672-711.
Figure 1. The structure of Amyloid beta A4 protein, peptide 672-711.
Creative Biostructure has rich professional experience in high quality Amyloid beta-sheet peptides production using detergent-free membrane protein expression system, we can perform various strategies for Mempro™ detergent-free protein production, including:
- Mempro™ Amyloid Beta-sheet Peptides Production Using Nanodiscs
Nanodisc is self-assembled soluble discoidal phospholipids bilayers, thus a functional stabilized membrane disk can be supplied by Nanodisc for the combination of membrane associated and membrane-bound molecules.
- Mempro™ Amyloid Beta-sheet Peptides Production Using Amphipols
Amphipols are a new class of surfactants, they are synthesized by forming amide bonds between octylamine and, sometimes sopropylamine. Amphipols are highly usable tools for handling membrane proteins in detergent-free water solutions and generally used for biophysical researches.
- Mempro™ Amyloid Beta-sheet Peptides Production Using Poly (styrene-co-maleic acid) Lipid Particles (SMALPs)
SMALPs are disc like structures including a protein in a lipid bilayer, parceled by the amphipathic styrene maleic acid polymer (SMA). As a biochemical approach, SMALP has a significant potential, for it makes membrane bilayers solubilized and it can implant discs in detergents-free condition.
These novel detergent-free approaches for Amyloid beta-sheet peptides production can be obtained easily, and enabling more comprehensively structural and functional studies.
Creative Biostructure provides other various Mempro™ membrane protein production services. Please feel free to contact us for a detailed quote.
References:
Roch, J. M. (1994). “Increase of synaptic density and memory retention by a peptide representing the trophic domain of the amyloid beta/A4 protein precursor”. Proceedings of the National Academy of Sciences 91(16), 7450-7454.
Buxbaum, J. D. (1993). “Protein phosphorylation inhibits production of Alzheimer amyloid beta/A4 peptide”. Proceedings of the National Academy of Sciences 90(19), 9195-9198.
Kubota, T. (2014). “Amyloid beta (A4) precursor protein expression in human periodontitis-affected gingival tissues”. Archives of oral biology 59(6), 586-594.
OPM, The structure of Amyloid beta A4 protein, peptide 672-711. November 2016 http://opm.phar.umich.edu/protein.php?pdbid=2lnq
