Based on the comprehensive protein engineering platform established through years of experiences, scientists form Creative Biostructure provide advanced custom Mempro™ C1 domain-containing protein production services using detergent-free expression system.
Cysteine-rich protein kinase C conserved region 1 (C1) domain is a phorbol esters/diacylglycerol binding domain, which can bind the lipid second messenger diacylglycerol (DAG) and the analogous phorbol esters. The binding of C1 domain and DAG is in a phospholipid and zinc-dependent manner. A number of signaling families have C1 domains, including chimaerins, RasGRPs, PKDs, diacylglycerol kinases (DGKs) and others. Conventional approaches for C1 domain-containing protein production require extraction from the membrane into detergent micelles to form detergent-solubilized complexes. However, detergent can cause conformational changes on the membrane proteins. Our alternative detergent-free expression system can best preserve the native conformation of C1 domain-containing proteins for further structural analysis.
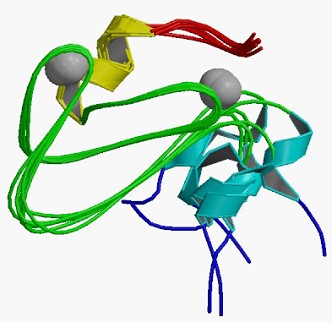
Figure 1. The structural model of C1 domain of Nore1. (Wikpedia)
Creative Biostructure has extensive expertise in high-yield C1 domain-containing protein production using detergent-free membrane protein expression system, we can perform various strategies for Mempro™ detergent-free C1 domain-containing protein production, including:
- Mempro™ C1 domain-containing protein production using nanodiscs
Nanodiscs are the self-assembly nanotechnology to stabilize membrane proteins removed from the membrane by membrane scaffold proteins (MSPs) in absence of detergent. Creative Biostructure can reconstitute C1 domain-containing proteins into nanodiscs in detergent-free methodology: Expressing the C1 domain-containing proteins in the cell-free expression system with the addition of pre-assembled nanodiscs.
- Mempro™ C1 domain-containing protein production using amphipols
The amphipathic amphipols have the ability to “trap” around transmembrane proteins, allowing them to stay folded. Creative Biostructure can apply A8-35 to solubilize C1 domain-containing protein during purification and stabilization processes.
- Mempro™ C1 domain-containing protein production using poly (styrene-co-maleic acid) lipid particles (SMALPs)
The SMALPs can be self-assembled by the simple addition of the SMA co-polymers. At neutral or alkaline pH, the disc-like structure assembles itself, encapsulating the C1 domain-containing proteins in a formation amenable to be purified.
These novel detergent-free techniques for membrane protein production can be obtained easily, and enabling more comprehensively structural and functional studies.
Creative Biostructure provides other various Mempro™ membrane protein production services. Please feel free to contact us for a detailed quote.
References:
C. Tribet, et al. (1996). Amphipols: Polymers that keep membrane proteins soluble in aqueous solutions. Proc Natl Acad Sci U. S. A. 93: 15047-15050.
F. Colón-González and M. G. Kazanietz (2006). C1 domains exposed: from diacylglycerol binding to protein-protein interactions. Biochim. Biophys. Acta., 1761(8): 827-837.
J. Das and G. M. Rahman (2014). C1 domains: structure and ligand-binding properties. Chem. Rev., 114 (24): 12108–12131.
M. Jamshad, et al. (2011). Surfactant-free purification of membrane proteins with intact native membrane environment. Biochem. Soc. Trans., 39: 813-818.
