Scientists from Creative Biostructure has built up a comprehensive protein engineering platform through years of experience. Creative Biostructure offers customized Mempro™ Glycolipid transfer proteins production services using detergent-free expression system.
Glycolipid transfer protein is responsible to transfer mainly glyceroglycolipids and glycosphingolipids between membranes in the cells, moreover, it is a cytosolic protein. It’s probably refer to translocation of glucosylceramides and found in brain, kidney, spleen, lung, cerebellum, liver and heart. The 3D structures of GLTPs from disparate species which have been determined are reveal a double-layered all-alpha-helical conformation. A ligand-binding site composed of a hydrophobic character for hydrocarbon chains and a sugar recognition center is contained in the conformation. This particular GLTP fold is different from other lipid transfer protein structures known by people. Generally, the structures of lipid transfer protein and protein bind lipids are dominated by β-sheet motifs, for example, β-cups, β-grooves and β-barrels.
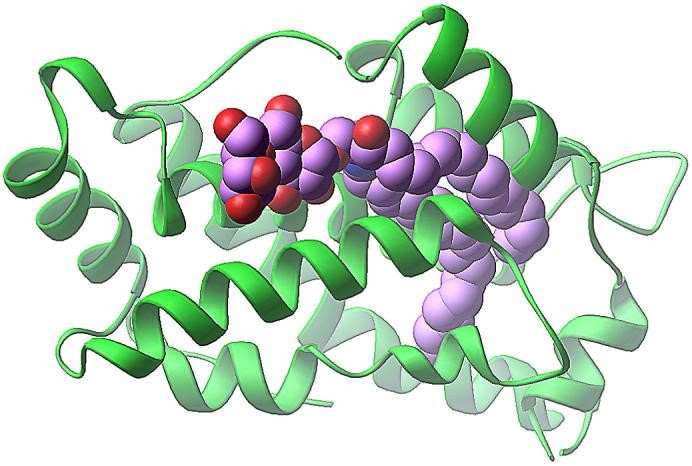
Figure 1. Crystal structure of the 18:1 LacCer-GLTP complex. (Biochim Biophys Acta. 2007)
Creative Biostructure has rich professional experience in high quality Glycolipid transfer proteins production using detergent-free membrane protein expression system, we can perform various approaches for Mempro™ detergent-free protein production, including:
- Mempro™ Glycolipid Transfer Protein Production Using Nanodiscs
Nanodiscs are soluble nanoscale phospholipid bilayers, which consist of membrane scaffold proteins (MSPs). MSPs supply two surface, one is a hydrophilic at the outside, while the other is facing the lipids. Nanodiscs possibly enable to maintain membrane proteins in solution, and offer a nature phospholipid bilayer environment in which the incorporated target keeps stably.
- Mempro™ Glycolipid Transfer Protein Production Using Amphipols
Using amphipols for structure determination of membrane proteins is to let membrane proteins water-soluble by means of trapping them with amphipathic polymers instead of detergents. Amphipols can be characteristically used in biophysics and biochemistry by the physicochemical properties of amphipols cut, which is based on the wide resources of polymer chemistry.
- Mempro™ Glycolipid Transfer Protein Production Using Poly (styrene-co-maleic acid) Lipid Particles (SMALPs)
This method generates SMALPs in which a little disk of lipid bilayer surrounded by polymer encircles the membrane proteins. Besides, the use of SMALPs can make detergent-free isolation of membrane proteins and retention of their crude lipid environment available.
These novel detergent-free technologies for glycolipid transfer proteins production can be obtained easily, and enabling more comprehensively structural and functional studies.
Creative Biostructure provides other various Mempro™ membrane protein production services. Please feel free to contact us for a detailed quote.
References:
Bayburt, T. H., & Sligar, S. G. (2010). Membrane protein assembly into Nanodiscs. FEBS letters, 584(9), 1721-1727.
Jamshad, M., Lin, Y.-P., Knowles, T. J., Parslow, R. A., Harris, C., Wheatley, M., . . . Overduin, M. (2011). Surfactant-free purification of membrane proteins with intact native membrane environment. Biochemical Society Transactions, 39(3), 813-818.
Prabudiansyah, I., Kusters, I., Caforio, A., & Driessen, A. J. (2015). Characterization of the annular lipid shell of the Sec translocon. Biochimica et Biophysica Acta (BBA)-Biomembranes, 1848(10), 2050-2056.
Tribet, C., Audebert, R., & Popot, J.-L. (1996). Amphipols: polymers that keep membrane proteins soluble in aqueous solutions. Proceedings of the National Academy of Sciences, 93(26), 15047-15050.
