Based on the comprehensive protein engineering platform established through years of experiences, scientists form Creative Biostructure can provide advanced custom Mempro™ integrin production services using detergent-free expression system.
Integrins are known as transmembrane proteins play important roles in cell-cell and cell-extracellular matrix (ECM) interactions. Integrins are actually the heterodimers, consisting of alpha and beta subunits. It has been proven that integrins aer identified in a variety of species, including mammals, chicken, zebrafish, etc. Notably, the integrin family in mankind has 24 members, which possess various combinations of 1 of 18 alpha- and 1 of 8 beta-subunits. Traditional techniques for transmembrane protein production require extraction from the membrane into detergent micelles. However, detergent can induce conformational changes on the transmembrane proteins. Our alternative detergent-free expression system can best preserve the native conformation of integrins for further structural analysis.
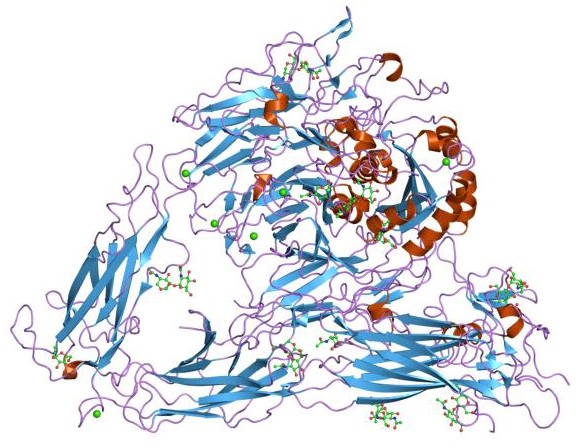
Figure 1. The structural model of the extracellular segment of integrin alpha Vbeta3. (Science, 2001)
Creative Biostructure has rich experience in high-quality integrin production in detergent-free membrane protein expression system, we can utilize various strategies for Mempro™ detergent-free integrin production, including:
- Mempro™ Integrin production using nanodiscs
Nanodiscs are the self-assembled nanotechnology to stabilize transmembrane proteins removed from the membrane by membrane scaffold proteins (MSPs) without detergent. Creative Biostructure can reconstitute integrins into nanodiscs in detergent-free procedure: Expressing the integrins in the cell-free expression system with the addition of pre-assembled nanodiscs.
- Mempro™ Integrin production using amphipols
The amphipathic amphipols have the ability to enwrap transmembrane proteins, allowing them to stay folded. Creative Biostructure can apply A8-35 to solubilize integrins during purification and stabilization processes.
- Mempro™ Integrin production using poly (styrene-co-maleic acid) lipid particles (SMALPs)
The SMALPs can be self-assembled by the simple addition of the SMA co-polymers. At neutral or alkaline pH, The disc-like structure assembles itself, encapsulating integrins in a formation amenable to be purified.
These novel detergent-free techniques for membrane protein production can be obtained easily, and enabling more comprehensively structural and functional studies.
Creative Biostructure provides other various Mempro™ membrane protein production services. Please feel free to contact us for a detailed quote.
References:
A. van der Flier and A. Sonnenberg (2001). Function and interactions of integrins. Cell Tissue Res., 305(3): 285-298.
C. Brakebusch and R. Fässler. (2003). The integrin-actin connection, an eternal love affair. EMBO J., 22(10):2324-2333.
C. Tribet, et al. (1996). Amphipols: Polymers that keep membrane proteins soluble in aqueous solutions. Proc Natl Acad Sci U. S. A. 93: 15047-15050.
J. P. Xiong, et al. (2001). Crystal structure of the extracellular segment of integrin alpha Vbeta3. Science, 294(5541): 339-345.
M. Jamshad, et al. (2011). Surfactant-free purification of membrane proteins with intact native membrane environment. Biochem. Soc. Trans., 39: 813-818.
