Creative Biostructure can provide unparalleled custom Mempro™ rhodopsin-like receptors and pumps production services using detergent-free expression system. You can count on us all through your projects.
Rhodopsin-like receptors and pumps , which are superfamily members of G-protein coupled receptors (GPCRs), are composed of 7 transmembrane helices (Figure 1). Rhodopsin-like receptors and pumps are extensively studied in biological function owing to their structural properties. Conventional approaches for rhodopsin purification need extraction from the membrane into detergent micelles to form detergent-solubilized samples. However, detergent can induce changes on membrane protein conformation. Stable solubilization and native conformation of rhodopsin is therefore an important issue in biotechnology as well as in biochemistry and structural biology. Our alternative detergent-free expression systems can best maintain the native conformation of rhodopsin-like receptors and pumps for further structural studies.
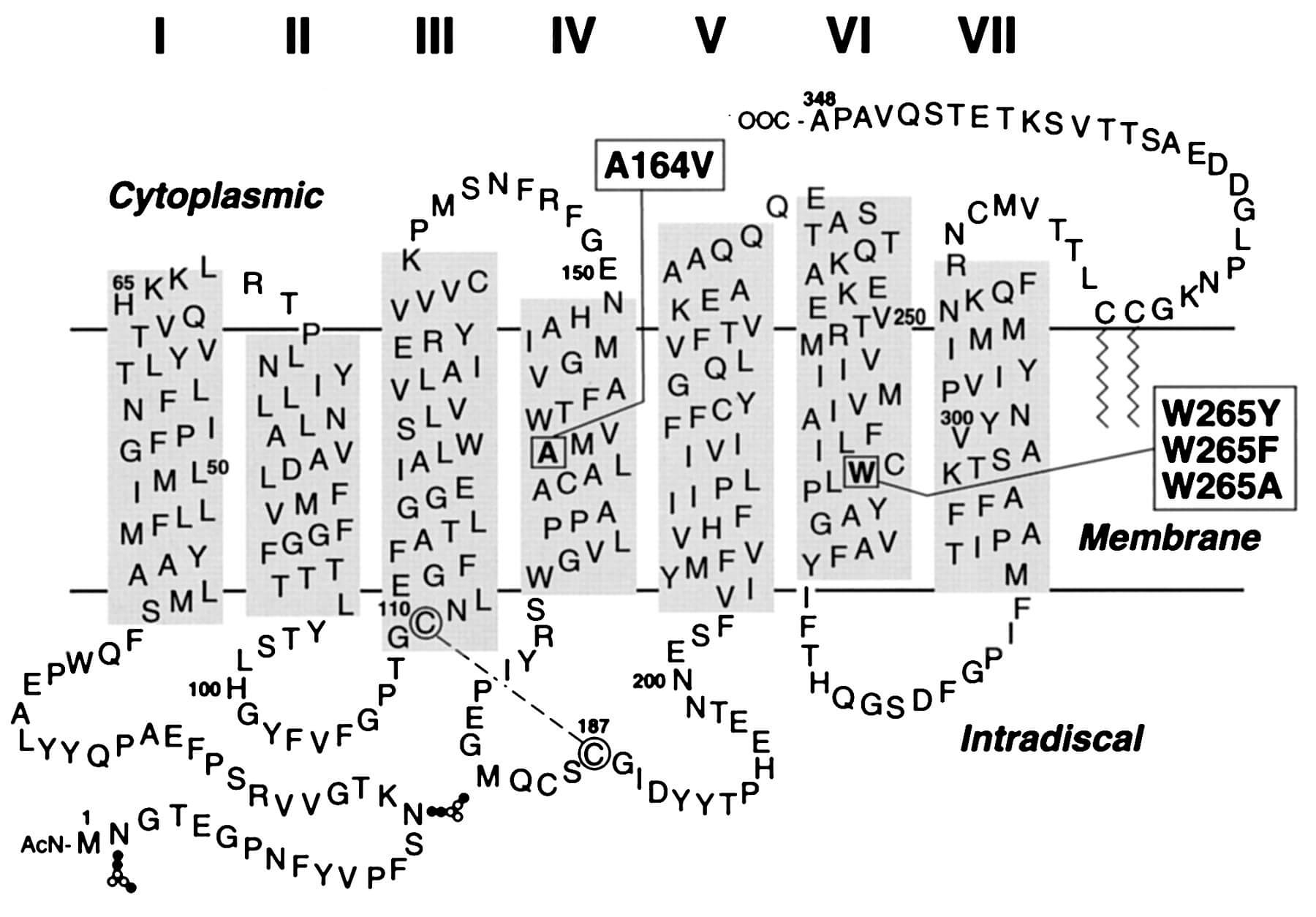 Figure 1. A secondary-structure model of bovine rhodopsin. (PNAS, 1999)
Figure 1. A secondary-structure model of bovine rhodopsin. (PNAS, 1999)
Specialists from Creative Biostructure have great expertise in high-yield rhodopsin-like receptors and pumps production using detergent-free membrane protein expression systmes, we can adopt various strategies for Mempro™ detergent-free rhodopsin-like receptors and pumps production, including:
- Mempro™ rhodopsin production using nanodiscs
Nanodiscs are the self-assembly system to stabilize membran proteins removed from the membrane by membrane scaffold proteins (MSPs) in no need of detergent. Creative Biostructure can reconstitute rhodopsin into nanodiscs in detergent-free methodology: Expressing the rhodopsin-like receptors and pumps in a cell-free system with the addition of pre-assembled nanodiscs.
- Mempro™ rhodopsin production using amphipols
The amphipathic amphipols have the ability to “trap” around transmembrane regions of proteins, allowing them to stay folded. Creative Biostructure can apply A8-35 to solubilize rhodopsin-like receptors and pumps during purification step.
- Mempro™ rhodopsin production using poly (styrene-co-maleic acid) lipid particles (SMALPs)
The SMALPs are self-assembled by the simple addition of the SMA co-polymer. At neutral or alkaline pH, a disc-like structure assembles itself, encapsulating the rhodopsin-like receptors and pumps in a form amenable to be purified.
These novel detergent-free technologies for rhodopsin-like receptors and pumps production can be obtained easily, and enabling more comprehensively structural and functional studies.
Creative Biostructure provides other various Mempro™ membrane protein production services. Please feel free to contact us for a detailed quote.
References:
J. P. Klare, et al. (2006). Effects of Solubilization on the Structure and Function of the Sensory Rhodopsin II/Transducer Complex. J. Mol. Biol., 356(5): 1207-1221.
P. J. Reeves, et al. (1999). Structure and function in rhodopsin: Kinetic studies of retinal binding to purified opsin mutants in defined phospholipid–detergent mixtures serve as probes of the retinal binding pocket. Proc. Natl. Acad. Sci. U. S. A., 96(5): 1927-1931.