With years of experience in the field of membrane protein production, Creative Biostructure now can provide high-quality custom Mempro™ FYVE/PHD zinc finger production service using virus-like particles.
The FYVE/PHD zinc finger superfamily contains three families: the FYVE family, the PHD domain family and the variant PHD-like domain family. The FTVE domain was identified and named after four proteins which are all Cys-rich proteins and contain this zinc finger domain: Fab1, YOTB, Vac1 and EEA1. FYVE domain is involved in membrane recruitment of cytosolic proteins because of its ability to bind to the phosphatidylinositol 3-phosphate (PI3P), which is the most ubiquitous phosphatidylinositide functions as a crucial cellular processes regulator. The PHD domain is identified based on a "Cys4-His-Cys3" zinc finger motif. The most important function of PHD domains is the recognition of epigenetic modification signals located on histones (histone H2A, H2B, H3 and H4), especially the tail regions of histones. The variant PHD-like domain family contains variant zinc finger domain members that share a similar motif with PHD domains, like RING zinc finger domains.
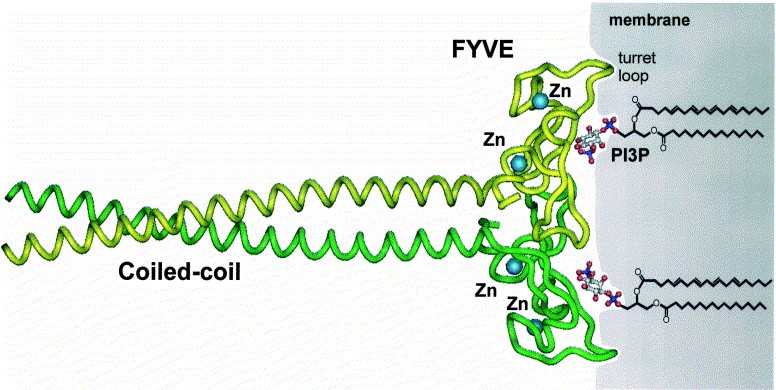 Fig. 1 Crystal structure of the EEA1 FYVE finger and adjacent coiled-coil region. (Harald Stenmark, et al. 2002)
Fig. 1 Crystal structure of the EEA1 FYVE finger and adjacent coiled-coil region. (Harald Stenmark, et al. 2002)
Virus-like particles (VLPs) can be described as viral "empty capsomere". It is a highly organized subunits result in the self-assembly of viral structural proteins. VLPs can mimic the native virus, except that they are all non-infectious because of lacking virus genetic materials. VLPs have been used in vaccine developments for a long time. The development of VLPs technology also provides a good choice for obtaining native or recombinant membrane proteins. VLPs can be obtained from various host systems including bacteria, yeasts, green plants, insect cells and mammalian cells, and many virus families have been reported to produce VLPs successfully including Retroviridae (e.g. HIV), bacteriophages (e.g. Qβ, AP205), Flaviviridae (e.g. Hepatitis C virus) and Parvoviridae (e.g. adeno-associated virus).
Creative Biostructure now can provide high yield, stable Mempro™ FYVE/PHD zinc finger production with our advanced VLP/lipoparticle technique. Various experimental systems are available in Creative Biostructure for customers to meet different requirements. We will co-express the viral core structural protein (e.g. retroviral structural core polyprotein Gag) along with the target FYVE/PHD zinc finger proteins or domains in host cells. The expressed core structural protein will proceed the self-assembly in the plasmid membrane, and bud-off from the plasmid membrane to form the lipoparticles (VLPs that that contain high concentration of specific membrane proteins within their construction), and the expressed target FYVE/PHD zinc finger proteins or domains shall be captured into the lipoparticles. The specific lipoparticles can be harvested at the surface of cells and the target proteins or domains can be isolated from the lipoparticles. The whole process is free from mechanical disruptions or detergents, so that the native structure and orientation of target inter-membrane proteins are protected perfectly.
Creative Biostructure can also provide a wide range of other Mempro™ membrane protein production service with various advanced techniques. Welcome to contact us for more details.
Reference:
Stenmark H, Aasland R, Driscoll P C. The phosphatidylinositol 3‐phosphate‐binding FYVE finger[J]. Febs Letters, 2002, 513(1): 77-84.
A.Roldão, et al. Virus-like particles in vaccine development.Expert Rev. Vaccines, 2010,9(10): 1149-1176.
Feussner I, Wasternack C. The lipoxygenase pathway [J]. Annual review of plant biology, 2002, 53(1): 275-297.
