Creative Biostructure offers specific custom Mempro™ lipocalin production services in virus-like particles system.
Virus-like particles (VLPs) mimic the native virus, which are non-infectious owing to they do not have any viral genetic materials. VLPs are self-assembly multiprotein structures, which are widely used for membrane protein production. By now, VLPs have been generated from components of a large variety of virus families including bacteriophages (e.g. Qβ, AP205), flaviviridae (e.g. Hepatitis C virus), retroviridae (e.g. HIV), and parvoviridae (e.g. adeno-associated virus). Virus-like particles carrying conformationally-complex proteins (termed lipoparticles) have been applied for integral membrane protein production. Lipoparticles can produce a large variety of structurally integrated membrane proteins, including G protein-coupled receptors (GPCRs), ion channels. Lipocalin proteins are a group of secreted proteins that transport lipids and other similar molecules such as steroids, bilins, and retinoids. The lipocalins share three short motifs that are the structurally conserved regions (SCRs), which are used to classify the lipocalin family into two main subfamilies, including the "kernel" and the "outlier" lipocalins. It has been shown that the kernel lipocalins possess all three SCRs while the outlier lipocalins contain only one or two of these SCRs. The lipocalins function as a potential marker for diagnosis and prognosis in several human diseases. Creative Biostructure can provide high-yield lipocalins production for biological and functional studies.
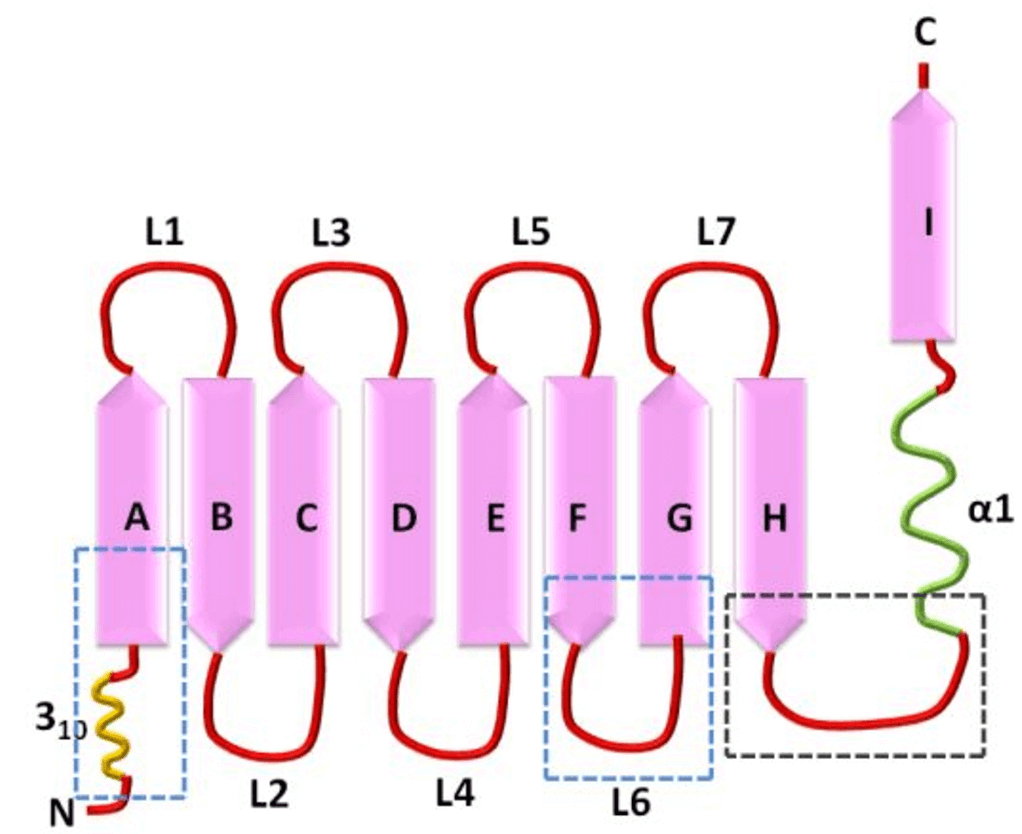 Figure 1. Schematitcs of the lipocalin fold. (Acute Phase Proteins-Regulation and Functions of Acute Phase Proteins, 2011)
Figure 1. Schematitcs of the lipocalin fold. (Acute Phase Proteins-Regulation and Functions of Acute Phase Proteins, 2011)
Creative Biostructure produces lipocalins in the stable and highly purified state in VLPs system. Lipoparticles can be generated from bacterial cells, yeast cells, insect cells, plant cells and mammalian cells. Well-characterized E. coli strains and insect cells are the most commonly used systems for VLPs production, as well as mammalian cells. For instance, we can obtain lipoparticles from mammalian cells by co-expressing the retroviral structural core polyprotein (termed Gag). Gag core proteins self-assemble at the plasma membrane, where they bud off and capture desired membrane proteins. Since lipocalins within lipoparticles are derived directly from the cell surface without mechanical disruption or detergents.
Creative Biostructure can also provide various custom Mempro™ membrane protein production services. Please feel free to contact us for a detailed quote.
References:
D. P. Patterson, et al. (2012). Virus-like particle nanoreactors: programmed encapsulation of the thermostable CelB glycosidase inside the P22 capsid. Soft Matter, 8: 10158-10166.
D. R. Flower (1996). The lipocalin protein family: structure and function. Biochem. J., 318(Pt 1): 1-14.
Lipocalin. (https://en.wikipedia.org/wiki/Lipocalin)
S. Chakraborty, et al. (2011). Neutrophil gelatinase associated lipocalin: structure, function and role in human pathogenesis. Acute Phase Proteins-Regulation and Functions of Acute Phase Proteins, Chapter 16, ISBN 978-953-307-252-4.
S. Willis, et al. (2008). Virus-like particles as quantitative probes of membrane protein interactions. Biochemistry, 47(27): 6988-6890.