Creative Biostructure can offer high-quality custom Mempro™ peptide fragments of transmembrane protein production services using virus-like particles system.
Virus-like particles (VLPs) can simulate the native virus, which are non-infectious due to they do not have any viral genetic materials. VLPs are self-assembly multiprotein structures, which are popularly used for integral membrane protein (transmembrane protein) production. Up to now, VLPs have been generated from components of a wide variety of virus families including bacteriophages (e.g. Qβ, AP205), Flaviviridae (e.g. Hepatitis C virus), Retroviridae (e.g. HIV), and Parvoviridae (e.g. adeno-associated virus). Recently, virus-like particles carrying conformationally-complex membrane proteins (termed lipoparticles) have been applied for integral membrane protein production. Lipoparticles can incorporate a wide range of structurally integrated membrane proteins, including G protein-coupled receptors (GPCRs), ion channels. Transmembrane protein (TM) is the protein spanning the entirety of the cell membrane, whic is embedded between the phospholipids. Transmembrane protein can be classified as alpha helical and beta barrel, depending on how the protein chain (also called peptide segments) is organized. Given that the characteristics of transmembrane protein, the accurate structural information of thes proteins is very difficut to be obtained. Therefore, Creative Biosturcture can produce peptide fragments of transmembrane proteins that providing information on their amino acid composition to facilitate the structural and functional of transmembrane proteins.
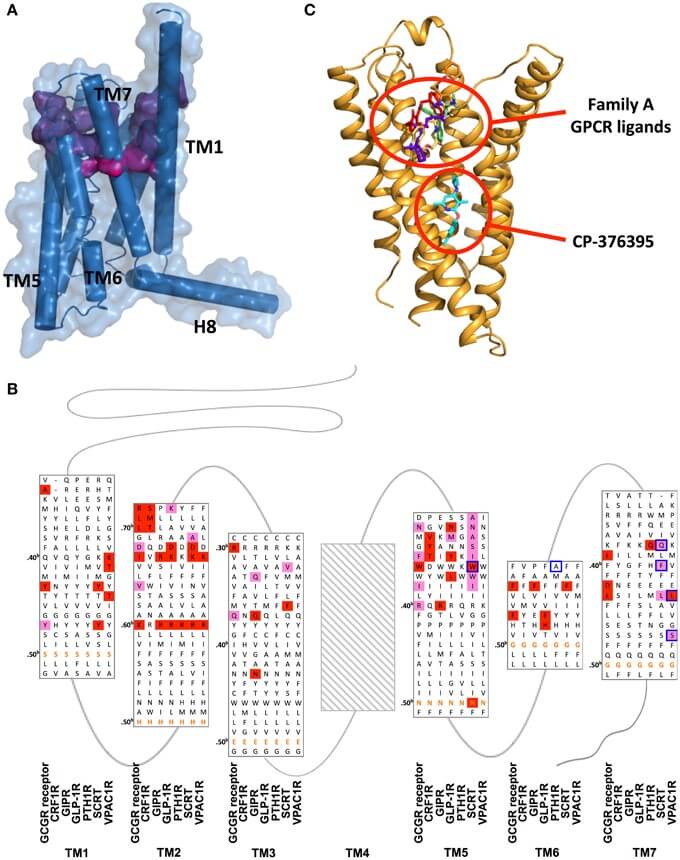
Figure 1. Ligand binding sites of family B GPCRs. (Front. Pharmacol, 2015)
Creative Biostructure generates peptide fragments of trnasmembrane proteins in the stable and highly purified state using VLPs system. Lipoparticles can be produced from bacterial cells, yeast cells, insect cells, plant cells and mammalian cells for peptide fragments of transmembrane proteins production. Well-characterized commercial Escherichia coli (E. coli) strains and insect cells are the most widely used systems for VLPs production, as well as mammalian cells. For instance, we can obtain lipoparticles from mammalian cells by co-expressing the retroviral structural core polyprotein (termed Gag). Gag core proteins self-assemble at the plasma membrane, where they bud off and capture target membrane proteins. Since the peptide fragments of transmembrane proteins within lipoparticles are derived directly from the cell surface without mechanical disruption or detergents.
Creative Biostructure provides other custom Mempro™ membrane protein production services. Please feel free to contact us for a detailed quote.
References:
A. Perdih, et al. (2012). Structural analysis of a peptide fragment of transmembrane transporter protein bilitranslocase. PLoS ONE, 7(6): e38967.
D. P. Patterson, et al. (2012). Virus-like particle nanoreactors: programmed encapsulation of the thermostable CelB glycosidase inside the P22 capsid. Soft Matter, 8: 10158-10166.
J. Venkatraman, et al. (2002). Structural analysis of synthetic peptide fragments from emre, a multidrug resistance protein, in a membrane-mimetic environment. Biochemistry, 41(21): 6631–6639.
K. J. Culhane, et al. (2015). Transmembrane signal transduction by peptide hormones via family B G protein-coupled receptors. Front. Pharmacol., 6: 264.
S. Willis, et al. (2008). Virus-like particles as quantitative probes of membrane protein interactions. Biochemistry, 47(27): 6988-6890.