Creative Biostructure has developed an advanced platform for membrane protein production, our scientists can provide custom Mempro™ CRAL-TRIO domain-containing protein production services using plant-based expression system.
Mempro™ plant-based protein production platform is a powerful system applied for production of membrane proteins. CRAL-TRIO domain belongs to a kind of protein structural domain that can bind small lipophilic ligands. The structure of CRAL-TRIO domain possesses several alpha helices as well as a beta sheet composed of six strands. It is reported that strands 2,3,4 and 5 can form a parallel beta sheet with strands 1 and 6 being anti-parallel. CRAL-TRIO domain is originally identified in GTPase-activating proteins (GAPs), guanine nucleotide exchange factors (GEFs) and hydrophobic ligand binding family proteins. In addition, CRAL binding protein carries 11-cis-retinol or 11-cis-retinaldehyde, while TRIO protein involves in coordinating actin remodeling, which is necessary for cell migration and growth.
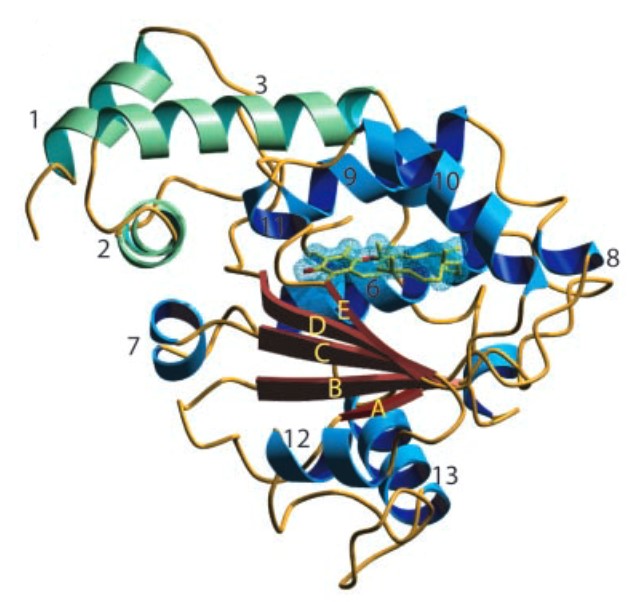 Figure 1. The secondary structure of human α-tocopherol (α-T) transfer protein (ATTP). (PNAS, 2003)
Figure 1. The secondary structure of human α-tocopherol (α-T) transfer protein (ATTP). (PNAS, 2003)
Creative Biostructure has rich professional experience in offering 1) animal-free, 2) low endotoxin, and 3) low protease level recombinant membrane proteins, which are particularly crucial for customers concerned with experimental variables caused by trace animal components or mammalian pathogens. Recombinant membrane proteins expressed by our plant system are free of animal components, serum, endotoxins and antibiotics as well as human or animal infectious agents or other endogenous mammalian contamination.
Of note, we can perform membrane protein production using various types of plants, such as Nicotiana benthamiana (tobacco), Medicago rativa (alfalfa), Arabidopsis thaliana (A. thaliana), potato, maize, barley and lettuce. Key features of Mempro™ plant-based expression system include:
1. Cost-effective2. Easy storage and distribution (barley grains containing desired recombinant proteins can be stored for many years)
3. Environment friendly (plants are cultivated in greenhouses)
4. Free of infectious or toxic contaminants
5. High scale-up capability
With the Mempro™ plant-based protein production platform, Creative Biostructure is capable of expressing, isolating, purifying and crystallizing CRAL-TRIO domain-containing proteins to facilitate the study of their structural and biological functions. Moreover, post-translational modification such as glycosylation and disulfide bonds can be introduced in our plant system.
Creative Biostructure can also provide Mempro™ plant-based virus-like particles (VLPs) production services, Mempro™ animal-free recombinant protein production services and Mempro™ membrane protein production services. Please feel free to contact us for a detailed quote.
References:
A. Wiktorek-Smagur, et al. (2012). Green way of biomedicine – how to force plants to produce new important proteins. Transgenic Plants-Advances and Limitations, Chapter 3. doi: 10.5772/1409.
J. M. bomar, et al. (2003). Mutations in a novel gene encoding a CRAL-TRIO domain cause human Cayman ataxia and ataxia/dystonia in the jittery mouse. Nature Gen., 35: 264-269.
K. Christopher Min, et al. (2003). Crystal structure of human α-tocopherol transfer protein bound to its ligand: Implications for ataxia with vitamin E deficiency. Proc. Natl. Acad. Sci. U.S.A.,100(25): 14713-14718.
K. G. Johnson and K. Kornfeld (2010). The CRAL/TRIO and GOLD domain protein TAP-1 regulates RAF-1 activation. Dev. Biol., 341(2): 464-471.
