Creative Biostructure can offer tailored Mempro™ plant-based pleckstrin homology (PH) domain-like protein production services. Our plant-derived expression system can best preserve the native conformation of PH domain-like proteins.
Mempro™ plant-derived protein production platform can produce animal-free, low endotoxin, and low protease activity membrane proteins. Animal-free recombinant membrane proteins are particularly crucial for customers concerned with experimental variables caused by trace animal components or mammalian pathogens. Notably, membrane proteins expressed by plant system are free of animal components, serum, endotoxins and antibiotics as well as human or animal infectious agents or other endogenous mammalian contamination. In addition, post-translational modification such as glycosylation and disulfide bonds can be introduced in plant system.
Creative Biostructure can offer membrane protein production services based on various types of plants, such as Nicotiana benthamiana (tobacco), Medicago rativa (alfalfa), Arabidopsis thaliana (A. thaliana), potato, maize, barley and lettuce. The significant advantages of plant-based membrane protein production include low cost, high yield, easy storage and distribution (such as barley grains containing desired recombinant proteins can be stored for many years), friendly environment (plants are usually cultivated in greenhouses), free of infectious or toxic contaminants, and high scale-up capability.
A large amount of signaling proteins possess a kind of small molecular domains (around 120 amino acids) that termed PH domains. It is reported that PH domain-like proteins have a beta-barrel topological structure, moreover, which is capped by an alph helix. The significant characteristics of PH domains is that this specific domains can introduce phosphoinositides phosphorylation at different sites within the inositol ringbind lipid. In other words, PH domains have lipid binding specificity.
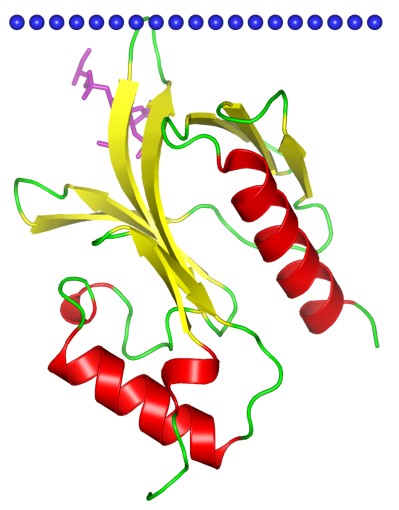 Figure 1. The structural model of PH domain of 3-phosphoinositide-dependent protein kinase 1. (OPM Database)
Figure 1. The structural model of PH domain of 3-phosphoinositide-dependent protein kinase 1. (OPM Database)
With the Mempro™ plant-based membrane protein production platform, Creative Biostructure is capable of expressing, isolating, purifying and crystallizing PH domain-like proteins to facilitate the study of their biological functions. Our renewed plant-based protein expression system can bypass numerous difficulties encountered in cell-based protein expression system, such as misfolding, aggregation, inactivity, poor stability and solubility, etc.
Creative Biostructure can also provide Mempro™ plant-based virus-like particles (VLPs) production services. Please feel free to contact us for a detailed quote.
References:
C. Pilling, et al. (2011). GRP1 PH domain, like AKT1 PH domain, possesses a sentry glutamate residue essential for specific targeting to plasma membrane PI(3,4,5)P3. Biochemistry, 50(45): 9845-9856.
F. Junge, et al. (2008) Large-scale production of functional membrane proteins. Cellular and molecular life sciences, 65 (11): 1729-1755.
M. A. Lemmon (2007). Pleckstrin homology (PH) domains and phosphoinositides. Biochem. Soc. Symp., 74: 81-93.