Creative Biostructure has professional experience in comprehensive membrane protein production, scientists from Creative Biostructure can offer customized Mempro™ chelatase production services using detergent-free expression system.
Magnesium-chelatase catalyses the primo step in the synthesis of bacteriochlorophy ll, the step is the insertion of Mg2+ into protoporphyrin IX. Magnesium-chelatase is a three-component enzyme and is regarded as an essential member in channeling intermediates into the chlorophyll branch in response to situations appropriate for photosynthetic growth. Protoporphyrin IX, ATP, Mg2+ and H2O are 4 substrates of this enzyme, however its 4 products are phosphate, ADP, Mg-protoporphyrin IX, and H+. This enzyme is one of the members of ligases, especially those forming nitrogen-D-metal bonds in coordination complexes. Magnesium-chelatase consists of three types of subunit, only one of them named Chll has a validated ATPase activity, while the ChlD subunit allosterically regulated the chelatase belongs to the AAA+ superfamily of ATPases. This superfamily combines ATP hydrolysis to various intracellular reactions, using the free energy of nucleotide hydrolysis to drive cellular processes and involving macromolecular remodeling.
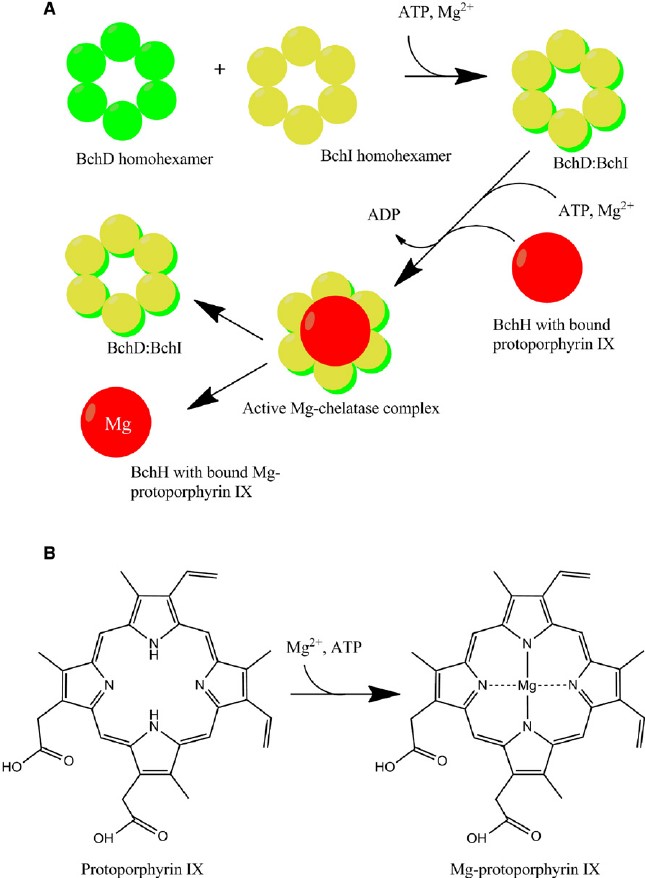 Figure 1. General pathway for catalysis by magnesium chelatase. (Structure, 2010)
Figure 1. General pathway for catalysis by magnesium chelatase. (Structure, 2010)
Creative Biostructure has rich experience in high-quality chelatase production using detergent-free membrane protein expression system, we can perform various strategies for Mempro™ detergent-free protein production, including:
- Mempro™ Chelatase Production Using Nanodiscs
Nanodiscs are assembled by a compound of detergent/phospholipid micelles and MSPs. Integral membrane proteins assemble themselves by nanodiscs, they are generally used for structural, enzymatic or biophysical researches.
- Mempro™ Chelatase Production Using Amphipols
Amphipols meaning small amphipathic polymers that are capable of holding individual MPs water-soluble in their native state under consist of little hydrophilic complexes. Amphipols are absolutely serviceable tools for handling membrane proteins in detergent-free aqueous solutions.
- Mempro™ Chelatase Production Using Poly (styrene-co-maleic acid) Lipid Particles (SMALPs)
SMALPs are disc in the lipid bilayer polymerized by styrene maleic acid copolymer (SMA). SMALPs cut discs from the cell membrane of nearly 11 nm in diameter. There are masses of obvious advantages by using SMALPs for structure determination.
With our incomparable detergent-free expression system and experience for hundreds of successful cases, Creative Biostructure guarantees our customers best service in chelatase production.
Creative Biostructure provides other various Mempro™ membrane protein production services. Please feel free to contact us for a detailed quote.
References:
Walker C J, Weinstein J D. In vitro assay of the chlorophyll biosynthetic enzyme Mg-chelatase: resolution of the activity into soluble and membrane-bound fractions[J]. Proceedings of the National Academy of Sciences, 1991, 88(13): 5789-5793.
WALKER J C, WILLOWS D R. Mechanism and regulation of Mg-chelatase[J]. Biochemical Journal, 1997, 327(2): 321-333.
Fodje M N, Hansson A, Hansson M, et al. Interplay between an AAA module and an integrin I domain may regulate the function of magnesium chelatase[J]. Journal of molecular biology, 2001, 311(1): 111-122.
