Based on the comprehensive membrane protein production platform established through years of experience, scientists from Creative Biostructure can provide tailored Mempro™ DNase I-like protein production services using detergent-free expression system.
DNase I-like protein is a superfamily which consists of three protein families: DNase I-like protein, Inositol polyphosphate 5-phosphatase (IPP5) and Sphingomyelin phosphodiesterase-like protein. IPP5 encoded by the ITPA gene in humans is one of the members of DNase I-like protein superfamily. However, in bacteria E. coli it’s encoded by the rdgB gene and in yeast S. cerevisiae is the HAM1 gene. Two dissimilar isoforms encoded by two transcript variants have already been discovered for the inosine triphosphate pyrophosphatase gene, meanwhile, more than two other transcript variants controlled protein-coding have been appraised. This enzyme has a multiple substrate-specificity and effects on other nucleotides, for example, deoxyxanthosine triphosphate and xanthosine triphosphate. The encoded protein serves as a homodimer which is discovered in HAM1 NTPase protein family.
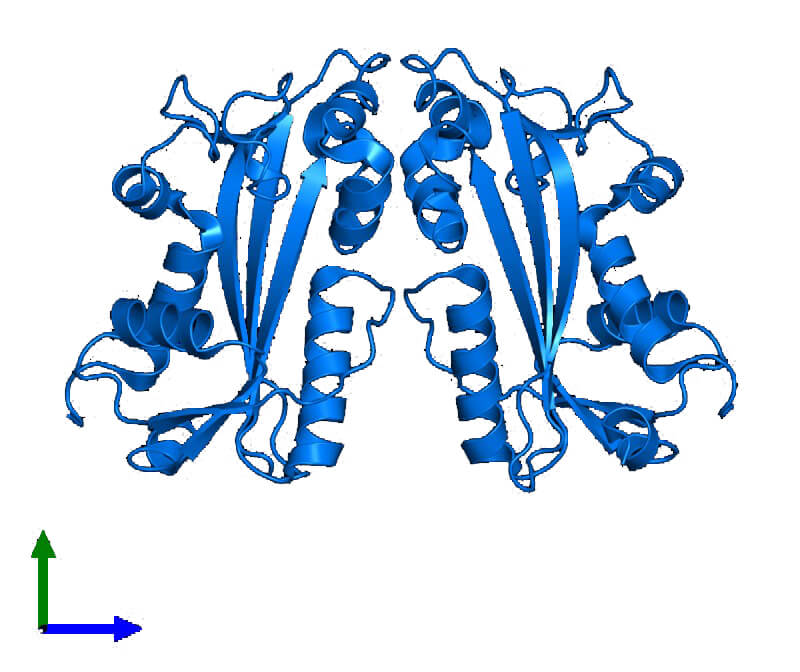
Figure 1. Crystal structure of inosine triphosphate pyrophosphatase protein. (J Biol Chem 2001)
Creative Biostructure has professional experience in high-quality DNase I-like protein production using detergent-free membrane protein expression system, we can perform various approaches for Mempro™ detergent-free protein production, including:
- Mempro™ DNase I-Like Protein Production Using Nanodiscs
Nanodiscs can be stabilized in solution through the assist of amphipathic helical scaffold protein, and also is an advanced tool to learn interacting companions of membrane embedded molecules. It has been indicated that Nanodiscs enable to deliver porphyrin-conjugated lipid in a fallow form. In addition, delivery of therapeutics is an amazing adhibition of the Nanodisc system.
- Mempro™ DNase I-Like Protein Production Using Amphipols
Using amphipols for structure determination of membrane proteins is to let membrane proteins water-soluble by means of trapping them with amphipathic polymers instead of detergents.
- Mempro™ DNase I-Like Protein Production Using Poly (styrene-co-maleic acid) Lipid Particles (SMALPs)
As a biochemical approach, SMALP has a significant potential for making membrane bilayers solubilized and implanting discs in detergents-free conditions. The comprehend of membrane proteins can possibly be advanced by biophysical studies of disc-structure.
These novel detergent-free approaches for DNase I-like protein production can be achieved easily, and enabling more comprehensively structural and functional studies.
Creative Biostructure provides other various Mempro™ membrane protein production services. Please feel free to contact us for a detailed quote.
References:
Lin, S., McLennan, A. G., Ying, K., Wang, Z., Gu, S., Jin, H., . . . Tang, R. (2001). Cloning, Expression, and Characterization of a Human Inosine Triphosphate Pyrophosphatase Encoded by the ITPAGene. Journal of Biological Chemistry, 276(22), 18695-18701.
Smith, M., Marinaki, A., Arenas, M., Shobowale‐Bakre, M., Lewis, C., Ansari, A., . . . Sanderson, J. (2009). Novel pharmacogenetic markers for treatment outcome in azathioprine‐treated inflammatory bowel disease. Alimentary pharmacology & therapeutics, 30(4), 375-384.
Jamshad, M., Lin, Y.-P., Knowles, T. J., Parslow, R. A., Harris, C., Wheatley, M., . . . Overduin, M. (2011). Surfactant-free purification of membrane proteins with intact native membrane environment. Biochemical Society Transactions, 39(3), 813-818.
Yan, N. (2015). Structural biology of the major facilitator superfamily transporters. Annual review of biophysics, 44, 257-283.
