Surface Plasmon Resonance (SPR)
Real-time analysis of biomolecular interactions and binding kinetics
Advanced Structural Biology Solutions Trusted by Leading Researchers
Creative Biostructure offers expert-driven, technology-focused, and cost-effective structural biology solutions to advance your research.
Structural biology is a field of life science that focuses on determining and analyzing the three-dimensional structures of biological molecules, particularly proteins, nucleic acids, and their complexes at atomic resolution.
This cross-cutting combination of biochemistry, biophysics, and computer algorithms allows researchers to probe the way that structural changes shape molecular function.
At Creative Biostructure, we provide an extensive portfolio of structural biology technologies to meet diverse research needs. Our expert team guides clients in selecting the most suitable techniques based on specific research objectives, sample characteristics, and resolution requirements.
Creative Biostructure offers multi-dimensional structural biology studies, integrating complementary technologies for deep insights into protein stability, conformational changes, and molecular recognition. Our expert team designs customized workflows combining biophysical methods to ensure reliable and comprehensive biomolecule characterization.
Real-time analysis of biomolecular interactions and binding kinetics
Direct measurement of binding thermodynamics and stoichiometry
Label-free detection of protein-protein and protein-ligand interactions
Solution-based measurement of molecular interactions with minimal sample consumption
High-throughput screening of small molecule binding to proteins
Quantitative analysis of protein thermal stability and folding characteristics
High-throughput screening of protein stability and ligand binding
Assessment of protein conformational stability under various conditions
Rapid assessment of protein secondary structure and conformational changes
Detailed analysis of protein secondary structure and aggregation states
Size distribution analysis and aggregation monitoring of proteins in solution
Absolute molecular weight determination and oligomeric state analysis
Label-free characterization of local protein structure and ligand binding
High-resolution analysis of protein size, shape, and complex formation
Analysis of protein oligomeric state and complex formation
Mapping protein dynamics and conformational changes with peptide-level resolution
Comprehensive characterization of antibody stability and functionality
We leverage advanced computational methods and AI technologies to predict, model, and analyze biomolecular structures and their dynamics, providing valuable insights for structural biology research.
- Structural Data Processing and Interpretation
- Structure Validation and Refinement
- Homology Modeling
- Ab Initio Structure Prediction
- AI-Based Protein Structure Prediction
- Molecular Dynamics (MD) Simulation
- Normal Mode Analysis (NMA)
- Principal Component Analysis (PCA)
- Protein-Protein Docking
- Molecular Docking
- Free Energy Calculations
- Quantum Mechanics/Molecular Mechanics (QM/MM)
Our structural biology expertise covers comprehensive structure determination and characterization of diverse biomolecules, from single-domain proteins to complex macromolecular assemblies, including soluble and membrane proteins, antibodies, nucleic acids, and their dynamic interactions in both stable and transient complexes.
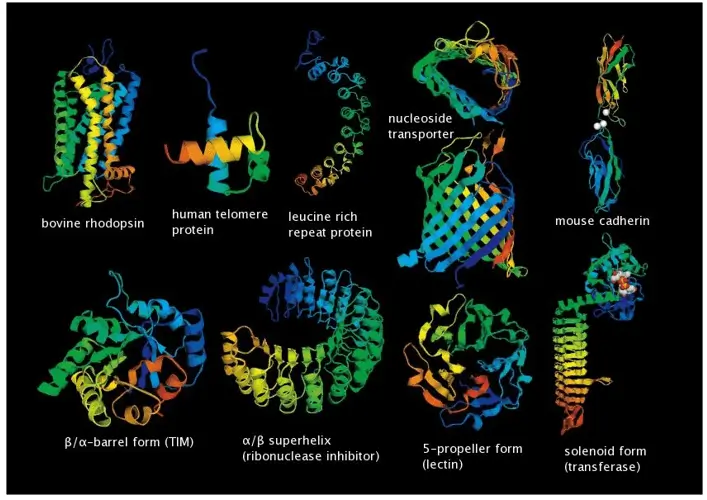 Various structures of proteins.
Various structures of proteins.
We have developed structural biology platforms including X-ray crystallography platform, NMR spectroscopy platform, electron microscopy (EM) platform, and we employ a variety of cutting-edge instruments to support our extensive macromolecular structure determination capabilities. Our facilities are equipped with crystallization robots for automated crystal screening, high-throughput crystal imaging systems, 300 kV Titan Krios G3i, Bruker AVANCE 800 MHz Spectrometer, etc. These state-of-the-art facilities, combined with our experienced team, ensure reliable and precise structural analysis for your research projects.
This project focuses on elucidating the three-dimensional structure of the Fab-antigen complex using X-ray crystallography. Our work begins with Fab preparation, followed by the formation of the Fab-antigen complex and initial crystal screening.

Figure 1. Protein crystal diffraction pattern.
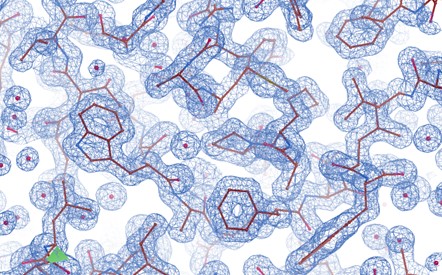
Figure 2. Density map of protein complex.
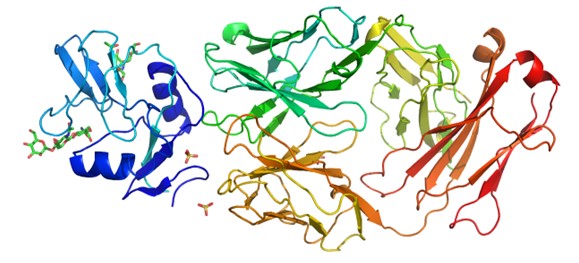
Figure 3. Protein structure of Fab-antigen complex.

Case Study
X-ray Crystallography for Fab-Antigen Complex Structural Analysis
Download this case study to discover the full analysis of Fab-Antigen complex using X-ray Crystallography, covering background, methods, results, and key insights.
Our unique combination of integrated technologies, customized solutions, expert team, and comprehensive support provides unparalleled structural biology insights, accelerating your research from basic characterization to applications.
Leverage advanced structural biology platforms, such as X-ray crystallography, NMR spectroscopy, and cryo-EM, for unparalleled insights.
Our team consists of highly experienced structural biologists, skilled in applying a range of technologies for precise biomolecular structure determination.
From consultation and project design to execution and data interpretation, we offer seamless, efficient services tailored to your specific needs.
We deliver high-precision protein structure services with competitive pricing and strategies that meet the unique requirements of your research.
Our structural biology services support drug discovery, protein engineering, and mechanism investigation, offering solutions for target structure analysis, protein-ligand interactions, structural-based drug design, and conformational dynamics studies.

Download our comprehensive brochure on structure-based drug discovery solutions to explore how our advanced techniques can accelerate your drug development process.
Here are a few key publications highlighting the impact of our structural biology services:
| Article | Journal | Year | Description |
|---|---|---|---|
| APOE Christchurch-mimetic therapeutic antibody reduces APOE-mediated toxicity and tau phosphorylation | Alzheimer's & Dementia | 2024 | …We resolved the three-dimensional crystal structure of purified 7C11.IgGFab protein as a fee-for-service with Creative Biostructure … |
| A novel mechanism of herbicide action through disruption of pyrimidine biosynthesis | Proceedings of the National Academy of Sciences | 2023 | … Creative Biostructure (Shirley, NY) provided the X-ray crystallographic support … |
| Linoleic acid binds to SARS-CoV-2 RdRp and represses replication of seasonal human coronavirus OC43 | Scientific Reports | 2022 | ...It is a complex structure of SARS-CoV-2 RNA polymerase with double-strand RNA fragments (Creative Biostructure, Shirley, NY) … |
| Synthesis and biological evaluation of selective survivin inhibitors derived from the MX-106 hydroxyquinoline scaffold | European Journal of Medicinal Chemistry | 2021 | … We are currently working with a CRO (Creative Biostructure, Inc) to optimize the crystallization condition in order to obtain the crystal structures of survivin protein in complexes with this … |
| All major cholesterol-dependent cytolysins use glycans as cellular receptors | Science advances | 2020 | … We thank Creative Biostructure (NY, USA) for protein chemical shift assignment … |
How do you handle membrane protein samples for structural studies?
We utilize a systematic workflow combining initial stability screening, detergent/lipid optimization, and structure determination primarily through Cryo-EM or X-ray crystallography, with comprehensive validation using complementary biophysical methods. In addition, we are skilled in reconstituting membrane proteins into nanodiscs, virus-like particles, and liposomes for downstream studies.
What sample requirements are needed for Cryo-EM analysis?
For optimal Cryo-EM analysis, we typically require samples at 0.5-1 mg/mL concentration with >90% purity, demonstrated homogeneity by SEC and DLS, and a minimum molecular weight of approximately 100 kDa.
What computational methods do you use for structure-based drug design?
Our comprehensive computational pipeline integrates virtual screening, molecular dynamics simulations, free energy calculations, and machine learning approaches, all validated with experimental binding data from SPR/ITC measurements.
What is your typical timeline for protein structure determination projects?
Project timelines vary based on complexity, but generally we deliver initial structures within 2-3 months for well-behaved proteins using X-ray crystallography or Cryo-EM.
How do you handle protein complexes that are challenging to reconstruct due to weak or transient interactions?
We employ a multi-technique approach combining crosslinking mass spectrometry (XL-MS), small-angle X-ray scattering (SAXS), and high-resolution Cryo-EM with advanced computational modeling to characterize challenging protein complexes with weak or transient interactions.
If you have any questions, please contact our support team, who are dedicated to helping you maximize the potential of your research with Creative Biostructure.
Related Resources
Ready to Advance Your Structural Biology Research?
Talk With An Expert →Contact our experts to discuss your structural analysis needs and receive a tailored quote for standalone or integrated solutions.
Online Inquiry
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.