Based on the comprehensive protein engineering platform established through years of experiences, scientists form Creative Biostructure can provide specific custom Mempro™ annexins production services using virus-like particles (VLPs). You can count on us all through your projects.
VLPs are self-assembled multiprotein structures, which can mimic the native virus, but are non-infectious due to they do not have any viral genetic information. VLPs derived from the Hepatitis B virus and consisted of the small HBV derived surface antigen (HBsAg). In addition, virus-like particles carrying native-conformation membrane proteins (termed lipoparticles) have been employed for the production of integral membrane protein. Lipoparticles can incorporate a wide range of structurally intact membrane proteins, including G protein-coupled receptors (GPCRs), and ion channels.
Annexins are a family of membrane binding proteins, which are Ca2+-regulated phospholipid binding proteins. Annexin family proteins contain four conserved structural elements termed annexin repeats consisting of approximately 70 amino acid residues. Notably, annexins can function as membrane scaffolding proteins to anchor other proteins to the cell membrane.
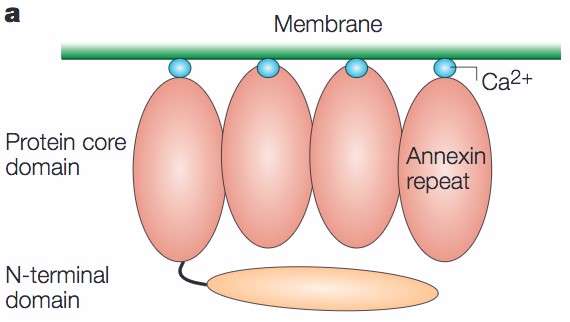 Figure 1. A schematic diagram of an annexin. (Nat. Rev. Mol. Cell Biol., 2005)
Figure 1. A schematic diagram of an annexin. (Nat. Rev. Mol. Cell Biol., 2005)
Creative Biostructure provides high-yield annexins in the stable, highly purified and native-conformation state. Lipoparticles can be generated from bacterial cells, yeast cells, insect cells, plant cells and mammalian cells for annexins production. Well-characterized commercial Escherichia coli (E. coli) strains and insect cells are the most widely used systems for VLPs production. Mammalian cells are also widely used for VLPs production with the target to construct vaccine candidates and gene therapy agents. For instance, lipoparticles can be obtained from mammalian cells by co-expressing the retroviral structural core polyprotein, Gag, along with a desired membrane protein. Gag core proteins self-assemble at the plasma membrane, where they bud off and capture target membrane proteins. Since the annexins within lipoparticles are derived directly from the cell surface without mechanical disruption or detergents, the native structure and orientation of annexins are retained.
Creative Biostructure provides other various Mempro™ membrane protein production services. Please feel free to contact us for a detailed quote.
References:
A. Roldão, et al. (2010). Virus-like particles in vaccine development. Expert Rev. Vaccines, 9(10): 1149-1176.
D. P. Patterson, et al. (2012). Virus-like particle nanoreactors: programmed encapsulation of the thermostable CelB glycosidase inside the P22 capsid. Soft Matter, 8: 10158-10166.
V. Gerke, et al. (2005). Annexins: linking Ca2+ signalling to membrane dynamics. Nat. Rev. Mol. Cell Biol., 6: 449-461.
U. Rescher and V. Gerke (2004). Annexins - unique membrane binding proteins with diverse functions. J. Cell Sci., 117: 2631-2639.
V. Gerke and S. E. Moss (2002). Annexins: from structure to function. Physiol. Rev., 82(2): 331-371.