Creative Biostructure can offer unparalled custom Mempro™ C2 domain-containing protein production services in virus-like particles system.
Virus-like particles (VLPs) mimic the native virus, which are non-infectious owing to they do not have any viral genetic materials. VLPs are self-assembly multiprotein structures, which are used for integral membrane protein production. So far, VLPs have been generated from components of a large variety of virus families including bacteriophages (e.g. Qβ, AP205), flaviviridae (e.g. Hepatitis C virus), retroviridae (e.g. HIV), and parvoviridae (e.g. adeno-associated virus). Recently, virus-like particles carrying conformationally-complex proteins (termed lipoparticles) have been applied for integral membrane protein production. Lipoparticles can incorporate a wide range of structurally integrated membrane proteins, including G protein-coupled receptors (GPCRs) and ion channels. C2 domain is a region that has a Ca2+-binding motif of around 130 residues in length. C2 domain can bind with enzymatic domains, for instance, phosphatase domain. The signifcant feature of C2 domain is that it has a wide range of lipid selectivity for the major components of eukaryotic cell membranes, including phosphatidylserine and phosphatidylcholine. Creative Biosturcture can produce C2 domain-containing proteins that providing information on their amino acid composition to facilitate the structural and functional of transmembrane proteins.
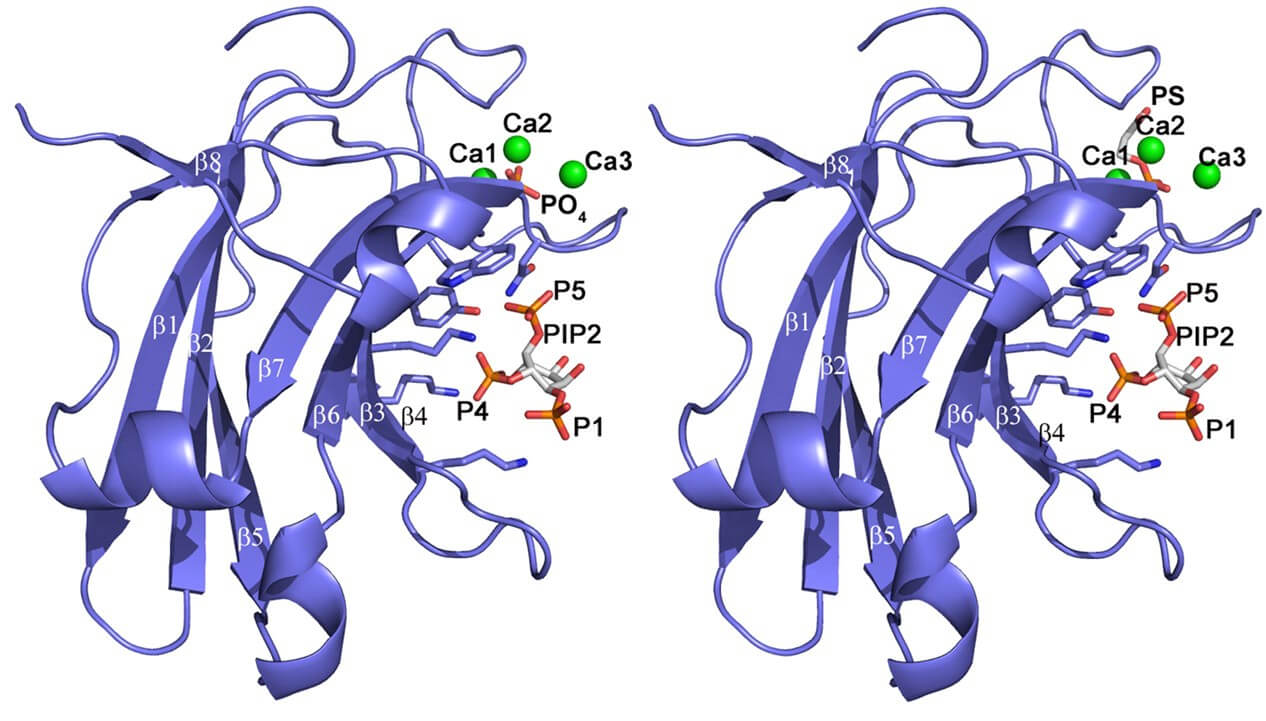 Figure 1. Structure of PKCα C2 domain bound to PtdIns(4,5)P2. (PNAS, 2009)
Figure 1. Structure of PKCα C2 domain bound to PtdIns(4,5)P2. (PNAS, 2009)
Creative Biostructure produces C2 domain-containing proteins in the stable and highly purified state in VLPs system. Lipoparticles can be generated from bacterial cells, yeast cells, insect cells, plant cells and mammalian cells for C2 domain-containing protein production. Well-characterized Escherichia coli (E. coli) strains and insect cells are the most commonly used tools for VLPs production, as well as mammalian cells. For example, we can obtain lipoparticles from mammalian cells by co-expressing the retroviral structural core polyprotein (termed Gag). Gag core proteins self-assemble at the plasma membrane, where they bud off and capture desired membrane proteins. Since the proteins containing C2 domain within lipoparticles are derived directly from the cell surface without mechanical disruption or detergents.
Creative Biostructure can also provide various custom Mempro™ membrane protein production services. Please feel free to contact us for a detailed quote.
References:
C2 domain. (https://en.wikipedia.org/wiki/C2_domain)
D. P. Patterson, et al. (2012). Virus-like particle nanoreactors: programmed encapsulation of the thermostable CelB glycosidase inside the P22 capsid. Soft Matter, 8: 10158-10166.
S. Willis, et al. (2008). Virus-like particles as quantitative probes of membrane protein interactions. Biochemistry, 47(27): 6988-6890.
M. Guerrero-Valero, et al. (2009). Structural and mechanistic insights into the association of PKCα-C2 domain to PtdIns(4,5)P2. Proc. Natl. Acad. Sci. U. S. A., 106(16): 6603-6607.
