Creative Biostructure has established a first-class platform for membrane protein production. Our custom Mempro™ membrane protein production of glycolipid transfer proteins is based on our leading cell-based expression systems, which perform the target membrane protein expression within a living cell.
Glycolipid transfer protein, also called GLTP for short, is a cytosolic protein. Glycolipid transfer protein can catalyse the glycolipids, but not the phospholipids or neutral lipids to transfer between different membranes. A lipid which needs to be transferred by an optimum GLTP transfer process should meet three structural requirements: one, the lipid must have two acyl chains; two, the lipid backbone must have an amide-nitrogen linked chain; three, the first saccharide must bind to the hydrophobic ceramide. Therefore, the lipid must be a glycosphingo-lipid. The glycolipids transfer of GLTP consists of several steps as follows: GLTP binds to the membranes, GLTP scans the membrane and uptakes the glycolipids, and then extracts the lipid from the intracellular membrane. GLTP transfers the glycosphingolipids, and this process play a pivot regulation function in various cell processes including cell growth, surface adhesion, and neurodevelopment.
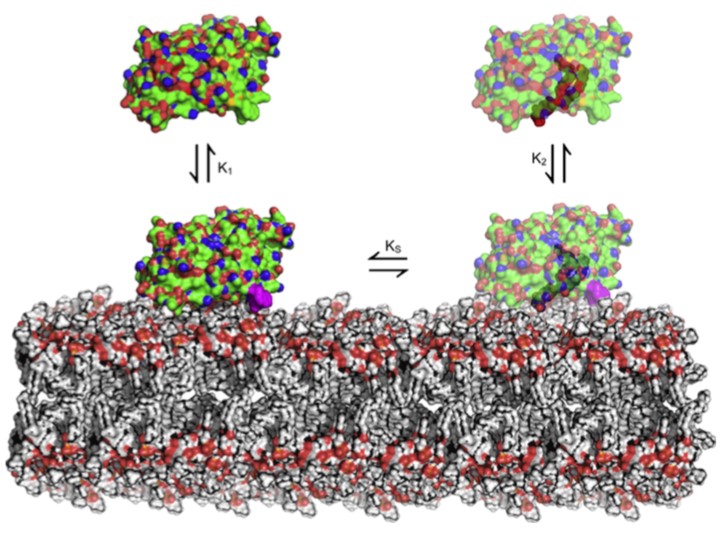
Figure 1.Schematic model of the ‘free transporter’ mechanism of GLTP. (Chemistry and Physics of Lipids. 2016)
Creative Biostructure can provide you the best membrane protein production services, and we can apply a lot of Mempro™ cell-based protein production systems including:
• Mempro™ Glycolipid Transfer Protein Production using Mammalian Cells System
Creative Biostructure can offer Mempro™ protein production in mammalian cells system. This system enables membrane proteins to perform correctly folding and post-translational modification. Applications for eukaryotic membrane protein production by Mempro™ mammalian cells system result in the best structural and functional characters.
• Mempro™ Glycolipid Transfer Protein Production in Yeast Cells System
Single-celled yeast has several advantages to be a culture host such as easy, quick and economic, and able to perform post-translational modification for eukaryotic membrane protein. Creative Biostructure has developed several strategies to improve yields per cell through optimizing the expression plasmid, host cell and culture conditions.
• Mempro™ Glycolipid Transfer Protein Production using Insect Cells System
Mempro™ protein production using insect cells system can not only increase the expression level but also reduce the truncated proteins compared to E. coli system, according to similar codon usage rules. Our innovational insect cells system based on baculovirus is advanced in several aspects as below:
• Accuracy;
• Easy scale-up;
• High membrane protein yield;
• Proper folding and post translation modifications;
• Use of cell lines ideal for suspension culture.
• Mempro™ Glycolipid Transfer Protein Production using Bacterial Cells System
Escherichia coli (E. coli) is the one of most widely applied bacterial host for membrane protein production. Optimized Lemo21 (DE3) strain is the culture host for our Mempro™ protein production in bacterial cells system. The target membrane protein encoding gene is located on a pET vector governed by the T7lac promoter. Membrane proteins are expressed as C-terminal GFP fusion protein as a result.
Creative Biostructure can conduct high-yield glycolipid transfer protein production by Mempro™ cell-based protein production services including expression, isolation, purification and crystallization.
We provide an array of Mempro™ membrane protein production services. Please feel free to contact us for a detailed quote.
References:
P. Mattjus. (2016). Specificity of the mammalian glycolipid transfer proteins. Chemistry and Physics of Lipids, 194: 72-78.
M. A. Kjellberg, et al. (2014). Alternation in the Glycolipid Transfer Protein Expression Causes Changes in the Cellular Lipidome. Plos One, 9(5): e97263.
V. R. Samygina, et al. (2011). Enhanced Selectivity for Sulfatide by Engineered Human Glycolipid Transfer Protein. Structure, 19(11): 1644–1654.