Creative Biostructure can provide unparalleled custom Mempro™ phosphatidylinositol 3-/4-kinase production services using cell-based expression system.
Mempro™ cell-based protein production system is popularly employed for the production of membrane proteins. It is reported that phosphatidylinositol (PI) metabolism plays an important role in various cellular functions in both animals and higher plants, including control of cell growth, regulation of cell cycle progression, a DNA damage checkpoint, recombination, and maintenance of telomere length. Phosphatidylinositol 3-kinase (PI3-kinase) is an enzyme that can phosphorylate phosphoinositides on the 3-hydroxyl group of the inositol ring and function as secondary messengers in cell signaling. Phosphatidylinositol 4-kinase (PI4-kinase) is an enzyme that can act on PI in the first committed step in the production of the secondary messenger inositol-1'4'5'-trisphosphate. PI 3-/4-kinases share a conserved catalytic domain at their C-terminal section.
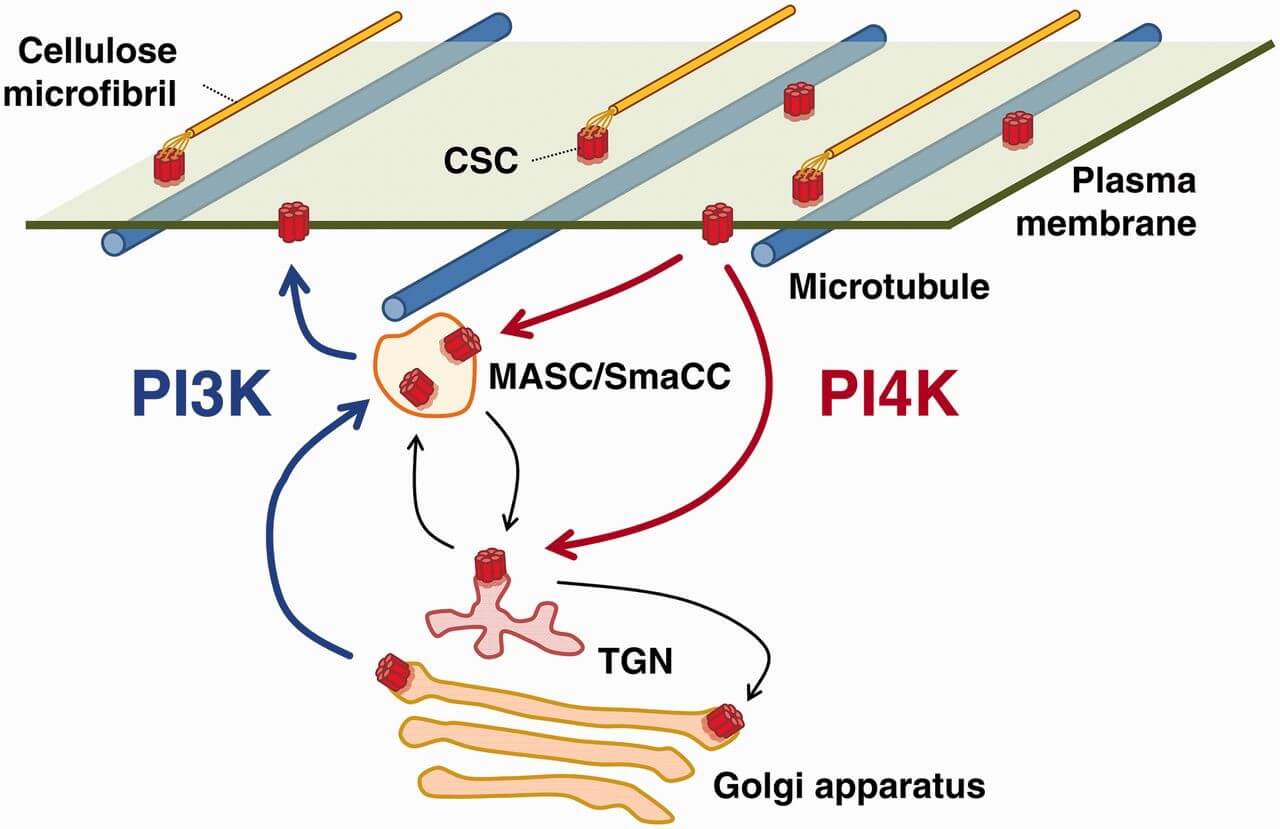 Figure 1. A schematic diagram of trafficking steps requiring PI4K and PI3K in CESA trafficking. (Plant Cell Phsiol., 2015)
Figure 1. A schematic diagram of trafficking steps requiring PI4K and PI3K in CESA trafficking. (Plant Cell Phsiol., 2015)
Creative Biostructure produces high-yield PI 3-/4-kinases using cell-based protein expression system, we can perform various strategies for Mempro™ cell-based protein production, including:
- Mempro™ Protein Production in Bacterial Cells System ;
- Mempro™ Protein Production in Yeast Cells System;
- Mempro™ Protein Production in Incest Cells System;
- Mempro™ Protein Production in Mammalian Cells System.
Escherichia coli (E. coli) is the most widely used bacterial host for the production of PI 3-/4-kinases. Yeast cells (such as saccharomyces cerevisiae and pichia pastoris) system, combines prokaryotic as well as eukaryotic characteristics. It is a powerful eukaryotic host owing to single cells, fast growth rates, inexpensive media, high cell densities. Insect cells (such as Sf9, Sf21 and High Five) are also the major system for PI 3-/4-kinases expression, which are developed from transfected insect cells in combination with vectors derived from the baculovirus species AcMNPV. Mammalian cells present another eukaryotic system for PI 3-/4-kinases expression, which provide near-native cell environment. Cell lines derived from COS, CHO, BHK-21, HEK293, HeLa and GH3 can be widely used for membrane protein expression.
With the Mempro™ cell-based protein production platform, Creative Biostructure is capable of expressing, isolating, purifying and crystallizing PI 3-/4-kinases to facilitate the study of their physiological functions.
We provide other various Mempro™ membrane protein production services. Please feel free to contact us for a detailed quote.
References:
C. Trometer, and P. Falson (2010). Mammalian membrane protein expression in baculovirus-infected insect cells. Methods Mol. Biol., 601: 105-117.
F. Junge, et al. (2008) Large-scale production of functional membrane proteins. Cellular Mol. Life Sci., 65 (11): 1729-1755.
J. Petschnigg, et al. (2011). Using yeast as a model to study membrane proteins. Curr. Opin. Nephrol. Hypertens., 20(4): 425-432.
J. Y. Jung, et al. (2002). Phosphatidylinositol 3- and 4-phosphate are required for normal stomatal movements. Plant Cell., 14(10): 2399-2412.
M. Fujimoto, et al. (2015). Phosphatidylinositol 3-kinase and 4-kinase have distinct roles in intracellular trafficking of cellulose synthase complexes in Arabidopsis thaliana. Plant Cell Physiol., 56(2): 287-298.