With the most advanced technology and years of experience in the field of membrane protein production, Creative Biostructure now provide Mempro™ custom cell-based phosphotyrosine protein phosphatases II production services for both academic and industrial uses.
The phosphotyrosine protein phosphatases II superfamily contains five membrane protein families, such as the mycobacterial PtpB-like family, the myotubularin-like phosphatases family, the higher-molecular-weight phosphotyrosine protein phosphatases family, the myo-inositol hexaphosphate phosphohydrolase (phytase) PhyA family and the dual specificity phosphatase-like family. Members of these families have been identified from many species such as human, Norway rat, mouse and Selenomonas ruminantium. These members are involved in the regulation of protein tyrosines (pTrys) phosphorylation. Protein Trys phosphorylation is an important post-translational modification (PTM) motif that provides a novel recognizing motif for protein interactions and cellular localisation. Through this way, phosphotyrosine protein phosphatases II superfamily can widely affect many cellular functions such as cell cycling and growth, proliferation, differentiation and transformation.
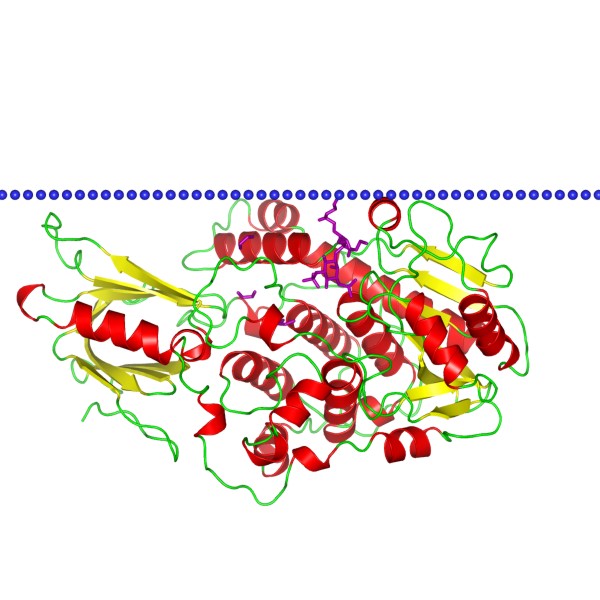 Figure 1. The schematic structure of myotubularin-related protein 2. (OPM Database)
Figure 1. The schematic structure of myotubularin-related protein 2. (OPM Database)
Cell-based membrane protein production system is the most widely applied technology in the field of membrane protein expression and purification. With the state-of-art Mempro™ cell based protein expression technique, Creative Biostructure now can provide our customers all kinds of Mempro™ cell-based protein expression systems for target phosphotyrosine protein phosphatases II protein production, including:
Mempro™ Protein Production in Bacterial Cells System;
Mempro™ Protein Production in Yeast Cells System;
Mempro™ Protein Production in Incest Cells System;
Mempro™ Protein Production in Mammalian Cells System.
Creative Biostructure can provide high-yield and high-quality custom Mempro™ phosphotyrosine protein phosphatases II production services. We will establish the optimal production strategy according to your purpose and special requirements. The bacteria systems (e.g. E. coli) are our common first choice. In bacteria expression systems, membrane proteins or their domains can be expressed as either soluble membrane protein or as inclusion bodies representing in cytoplasmic aggregates. The yeast cell systems combine many advantages of both prokaryotic and eukaryotic systems such as relative economic costs, single cells, high cell densities, faster grow rates, etc. Insect cell systems such as Sf9, Sf21 and High Five are also applied in many cases. These insect cell systems are developed from transfected cells of insects and combined with vectors derived from the baculovirus species AcMNPV. Mammalian cell expression systems provide nearly-native intracellular environments for target membrane protein production, which make them the most powerful technique. Cell lines derived from HEK293, BHK-21, CHO, COS, HeLa and GH3 are widely applied for integral membrane protein production. We will take every detailed characteristic and your requirements into our consideration to establish the optimal strategy in each individual case. With our Mempro™ services,your membrane protein production process will be easier.
Creative Biostructure also provides a wide range of other custom membrane protein production services with multiple advanced technologies. Welcome to contact us for more details.
References:
Morona J K, Morona R, Miller D C, et al. Streptococcus pneumoniae capsule biosynthesis protein CpsB is a novel manganese-dependent phosphotyrosine-protein phosphatase[J]. Journal of bacteriology, 2002, 184(2): 577-583.
Caselli A, Chiarugi P, Camici G, et al. In vivo inactivation of phosphotyrosine protein phosphatases by nitric oxide[J]. FEBS letters, 1995, 374(2): 249-252.
C. Trometer, and P. Falson (2010). Mammalian membrane protein expression in baculovirus-infected insect cells. Methods Mol. Biol., 601: 105-117.
S. Schlegel, et al. (2013). Bacterial-based membrane protein production. Biochim. Biophys. Acta., 1843(8): 1739-1749.