Based on the comprehensive protein engineering platform through years of experience, experts from Creative Biostructure provide customized Mempro™ glycolipid transfer protein production using advanced virus-like particles technique.
Glycolipid transfer proteins (GLTP) are defined by their abilities to catalyze the intermembrane transfer of glycolipids between different intercellular membranes. GLTPs are small (about 24kD), soluble cytosolic proteins and are ubiquitous in brain, kidney, spleen, lung, cerebellum, liver and heart. GLTP obviously differs from other glycolipid-binding proteins such as nonspecific lipid transfer proteins, lectins, lung surfactant proteins, lysosomal sphingolipid activator proteins, etc. GLTPs can specially bind with both glycerol- and sphingoid-based glycolipids, but its specificity critically depends upon that the initial sugar residue be beta-linked to the non-polar, hydrophobic lipid backbone. 3D protein structures of GLTPs have indicated a unique lipid binding mode and a unique conformational architecture that targets GLTP to membranes.
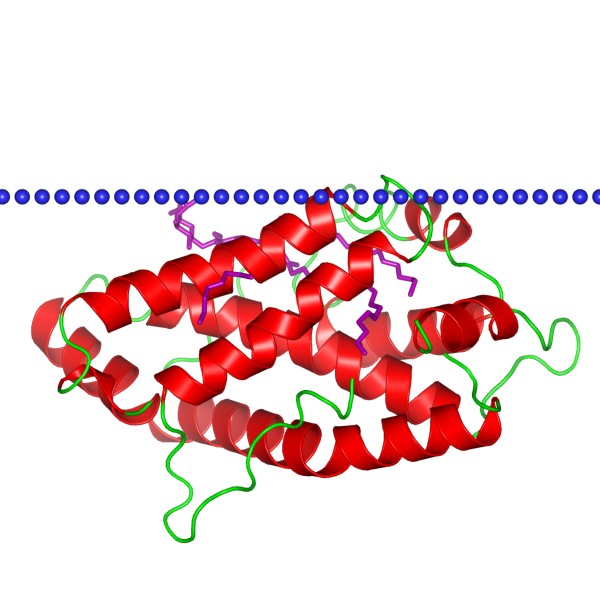 Figure 1. The schematic structure of Glycolipid transfer protein, conformation 1 (OPM Database)
Figure 1. The schematic structure of Glycolipid transfer protein, conformation 1 (OPM Database)
Virus-like particles (VLPs), define as viral "empty shells" which are highly organized subunits formed by viral structural proteins. VLPs have been widely used in vaccine development, antigen delivery and other biological or bio-medical programs for a quite long time. Many virus families have been reported to successfully produce VLPs such as Parvoviridae, bacteriophages, Retroviridae, etc. VLPs now can be obtained from various host systems including bacteria, yeasts, insect cells, mammalian cells.
Lipoparticles are VLPs that contain high concentration of specific membrane proteins within their construction. As reported by many purification researches, lipoparticles can be used for recombinant membrane protein productions with several advantages. Higher yield and stability can be achieved by using VLPs/lipoparticles technique. Target membrane proteins can be protected by lipoparticles during the isolation and purification. The orientation of membrane proteins and structure can remain native and complete during the isolation.
With years of experience in membrane protein production, Creative Biostructure now can provide high-quality custom Mempro™ glycolipid transfer protein production using advanced virus-like particles technique. Multiple experimental systems are available for customers to meet different requirements in your cases. We will establish the optimal strategies for you with our advanced techniques. Co-expressing of viral core structural protein and target GLTP protein in the host cells will be achieved to obtain the specific lipoparticles. The expressed viral core structural protein will perform the self-assembly within the plasmid membrane, and budding-off from the plasmid membrane to form the lipoparticles. During the same time the expressed target GLTP proteins shall be captured within the lipoparticles, and transformed to the outside of the host cells along with the lipoparticles. We will harvest the lipoparticles at the surface of host cells and obtain the target GLTP protein. The entire process is free from detergents and mechanical disruptions, and the target GLTP protein will be protected perfectly by lipoparticles.
Creative Biostructure also provides various other Mempro™ membrane protein production services using our advanced VLPs technique. Welcome to contact us for more details.
Reference:
A.Roldão, et al. Virus-like particles in vaccine development.Expert Rev. Vaccines, 2010,9(10): 1149-1176.
Sasaki T. Glycolipid transfer protein and intracellular traffic of glucosylceramide[J]. Experientia, 1990, 46(6): 611-616.
Brown R E, Mattjus P. Glycolipid transfer proteins[J]. Biochimica et Biophysica Acta (BBA)-Molecular and Cell Biology of Lipids, 2007, 1771(6): 746-760.
