Creative Biostructure has established Mempro™ membrane protein production platform through years of experience, we ensure producing high-quality saposins using plant-derived expression system.
Creative Biostructure is knowledgable in providing unmatched membrane protein production services based on various types of plant hosts, such as Nicotiana benthamiana (tobacco), Medicago rativa (alfalfa), Arabidopsis thaliana (A. thaliana), potato, maize, barley and lettuce. We can perform two different strategies for recombinant membrane protein production in plants: transient expression and stable transformation.
Mempro™ plant-based membrane protein production platform has lots of characteristics, such as animal-free, low protease activity, and low endotoxin. Animal-free recombinant membrane proteins are very important for customers concerned with experimental variables caused by trace animal components or mammalian pathogens. Recombinant membrane proteins expressed by our plant system are free of animal components, serum, endotoxins and antibiotics as well as human or animal infectious agents or other endogenous mammalian contamination. Additionally, post-translational modification such as glycosylation and disulfide bonds can be introduced in plant system.
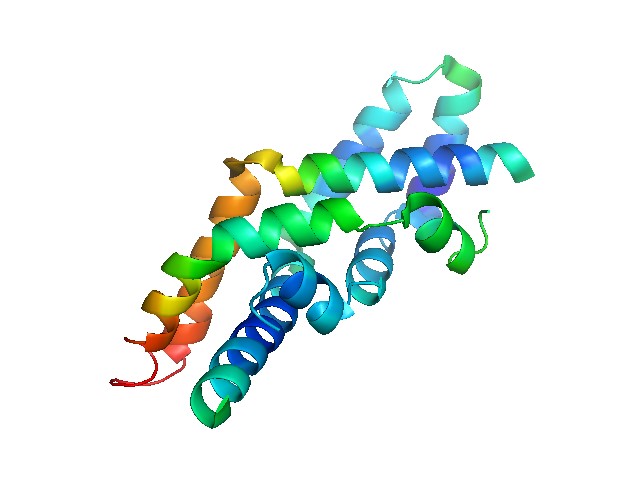 Figure 1. Crystal structure of human saposin C dimer in an open conformation. (Wikipedia)
Figure 1. Crystal structure of human saposin C dimer in an open conformation. (Wikipedia)
Saposins, also called sphingolipid activator proteins (SAPs), belong to small glycoproteins activating lysosomal hydrolases that participate in metabolism of sphingolipids. Sphingolipids are involved in the regulation of various cellular functions, such as growth, differentiation and apoptosis. Sphinogolipids are degraded in the lysosome, and the products from their hydrolysis are used in other biosynthetic and regulatory pathways in the host. There are a number of lysosomal enzymes involved in the breakdown of sphinogolipids, and these act in sequence to degrade the moieties. Saposins are non-enzymatic and usually have a low molecular weight. They are conserved across a wide range of eukaryotes and contain specific saposin domains that aid in the activation of hydrolase enzymes. There have been four human saposins described, sharing significant similarity with each other and with other eukaryotic SAP proteins.
With the Mempro™ plant-based protein production platform, Creative Biostructure is capable of expressing, isolating and purifying saposins to facilitate the study of their biological functions. We can overcome the common difficulties encountered in cell-based expression system, such as misfolding, aggregation, inactivity, high endotoxin, poor stability and solubility, etc.
Creative Biostructure can also provide Mempro™ plant-based virus-like particles (VLPs) production services, Mempro™ animal-free recombinant protein production services and Mempro™ membrane protein production services. Please feel free to contact us for a detailed quote.
References:
A. Wiktorek-Smagur, et al. (2012). Green way of biomedicine – how to force plants to produce new important proteins. Transgenic Plants-Advances and Limitations, Chapter 3. doi: 10.5772/1409.
Saposin (IPR008373). InterPro. (https://www.ebi.ac.uk/interpro/entry/IPR008373)
Y. Kishimoto, et al. (1992). Saposins: structure, function, distribution, and molecular genetics. J. Lipid Research, 33(9): 1255-1267.
