Creative Biostructure can provide unmatched custom Mempro™ single-helix ATPase regulators production services based on virus-like particles (VLPs) system.
Single-helix ATPase regulators are a large group of the bitopic proteins with alpha-helical transmembrane anchors and can modulate ATPases. It is reported that alpha-helices and beta-barrels are the main components of the transmembrane domains in transmembrane proteins. Single-helix ATPase regulators can be classified two families: calcium ATPase regulators and FXYD regulators. Calcium ATPase regulators locate in endoplasm reticulum (ER) and regulate calcium homeostasis. Members of calcium ATPase regulators family include phospholamban, sarcolipin and cytoplasmic helix. FXYD regulators locate in the plasma membrane of eukaryo, members of FXYD regulators family are tissue-specific modulators of Na, K-ATPase. FXYD1 mainly increases the apparent affinity for intracellular Na+ of Na, K-ATPase. FXYD2 and FXYD4 can oppositely regulate the apparent affinity for Na+ of Na, K-ATPase.
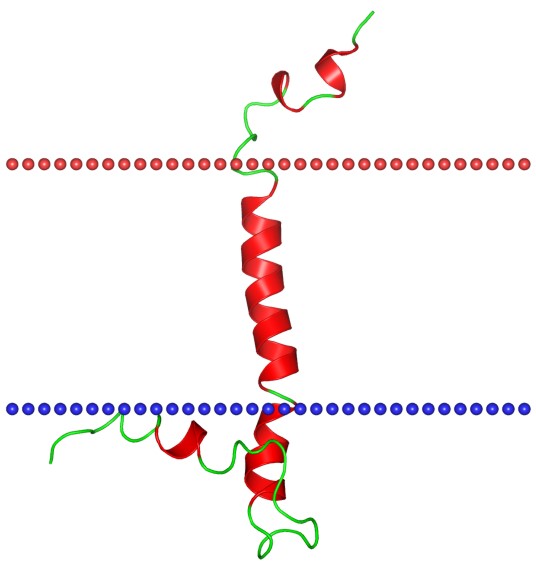
Figure 1. The structural model of Na,K-ATPase regulatory protein FXYD1 (phospholemman). (Wikipedia)
Virus-like particles (VLPs), are self-assembled multiprotein structures, can mimic the native virus. However, VLPs are non-infectious owing to lack of any viral genetic materials. It is reported that VLPs are obtained from the Hepatitis B virus and consist of the small HBV derived surface antigen (HBsAg). Currently, virus-like particles carrying conformationally-complex membrane proteins (termed lipoparticles) have been used for transmembrane protein production. Lipoparticles can incorporate a wide range of structurally intact membrane proteins, including G protein-coupled receptors (GPCRs), ion channels.
Creative Biostructure provides high quality single-helix ATPase regulators in the stable, highly purified and native-conformation state. Lipoparticles can be produced from bacterial cells, yeast cells, insect cells, plant cells and mammalian cells for single-helix ATPase regulators production. Escherichia coli (E. coli) strains and insect cells are the most popularly used systems for VLPs production. Mammalian cells are also widely employed for VLPs production with the target to construct vaccine candidates. For instance, we can obtain lipoparticles from mammalian cells by co-expressing the retroviral structural core polyprotein, Gag, along with the desired transmembrane protein. Gag core proteins self-assemble at the plasma membrane, where they bud off and capture target membrane proteins. Since the single-helix ATPase regulators within lipoparticles are derived directly from the cell surface without mechanical disruption or detergents, the native structure and orientation of transmembrane proteins are retained.
Creative Biostructure also provides other various Mempro™ membrane protein production services. Please feel free to contact us for a detailed quote.
References:
A. Roldão, et al. (2010). Virus-like particles in vaccine development. Expert Rev. Vaccines, 9(10): 1149-1176.
D. H. MacLennan and E. G. Kranias. (2003) Phospholamban: a crucial regulator of cardiac contractility. Nat. Rev. Mol. Cell Biol., 4(7): 566-77.
K. Geering (2005). Function of FXYD proteins, regulators of Na, K-ATPase. J. Bioenerg. Biomembr., 37(6): 387-92.
S. Willis, et al. (2008). Virus-like particles as quantitative probes of membrane protein interactions. Biochemistry, 47(27): 6988-6890.
