Based on the comprehensive protein engineering platform established through years of experience, scientists from Creative Biostructure offer tailored Mempro™ transmembrane proteins with NAD (P)-binding Rossmann-fold domains production services using detergent-free expression system.
Transmembrane proteins with NAD (P)-binding Rossmann-fold domains are made up of two protein families: tyrosine-dependent oxidoreductases and flavin-containing amine oxidoreductase. The tyrosine-dependent oxidoreductases family contains 8 members and the flavin-containing amine oxidoreductase family has 6 members, all of the 14 proteins are shown in the following table. MAOB is one of the members of the flavin monoamine oxidase family and is encoded by the MAOB gene in human, moreover, it is located in the outer mitochondrial membrane. Phenylethylamine and benzylamine are degraded by MAOB, while MAOB plays a crucial role in the catabolism of neuroactive and vasoactive amines.
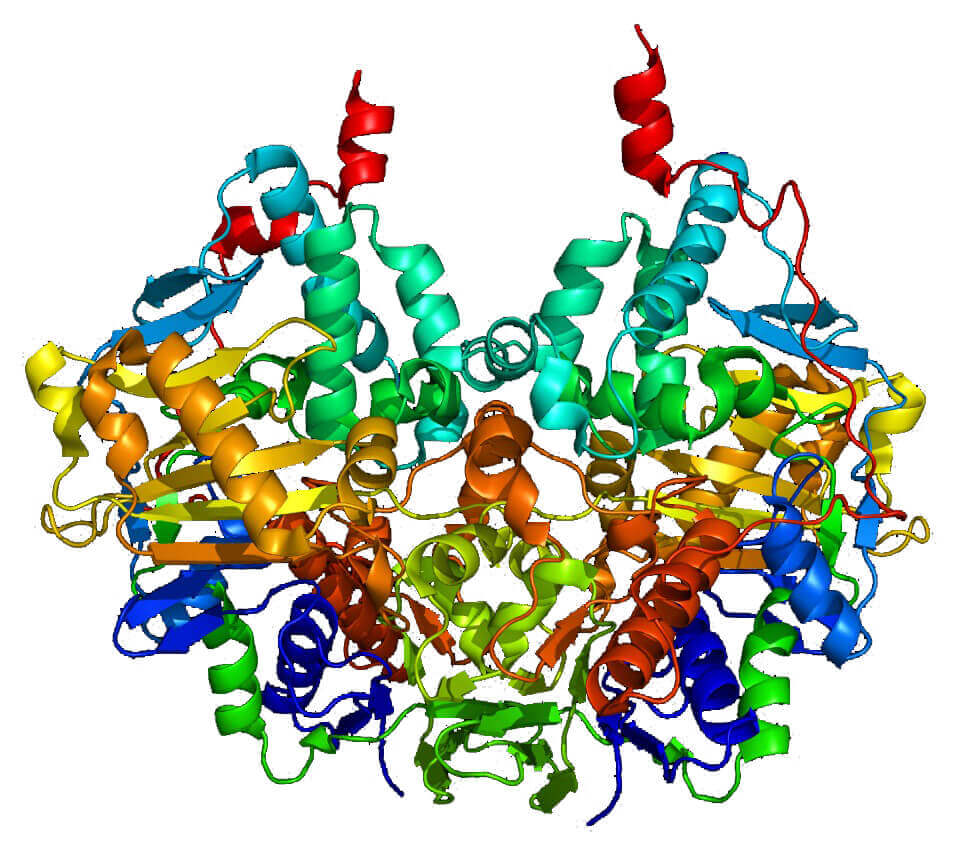 Figure 1. The structure of MAOB protein. (Arch. Biochem. Biophys 2007)
Figure 1. The structure of MAOB protein. (Arch. Biochem. Biophys 2007)
| Family | Protein Name | Species | Localization |
| Flavin containing amine oxidoreductase | Monoamine oxidase B | Homo sapiens | Mitochon. outer |
| Monoamine oxidase A | Rattus norvegicus | Mitochon. outer | |
| Monoamine oxidase B | Rattus norvegicus | Mitochon. outer | |
| Protoporphyrinogen oxidase | Nicotiana tabacum | Mitochon. inner | |
| Monoamine oxidase A | Homo sapiens | Mitochon. outer | |
| Alpha-glycerophosphate oxidase (GlpO) | Streptomyces sp. | Bact. Gram-pos plas | |
| Protoporphyrinogen oxidase | Homo sapiens | Mitochon. inner | |
| Protoporphyrinogen oxidase | Myxococcus xanthus | Bact. Gram-neg inner | |
| Tyrosine-dependent oxidoreductases | Corticosteroid 11-beta-dehydrogenase, isozyme 1 | Mus musculus | Endoplasm. reticulum |
| Corticosteroid 11-beta-dehydrogenase, isozyme 1 | Homo sapiens | Endoplasm. reticulum | |
| Corticosteroid 11-beta-dehydrogenase, isozyme 1 | Cavia porcellus | Endoplasm. reticulum | |
| Tropinone reductase 2 | Datura stramonium | Eukaryo. plasma | |
| Sorbitol dehydrogenase | Rhodobacter sphaeroides | Bact. Gram-neg inner | |
| Pteridine reductase 1 | Leishmania tarentolae | Eukaryo. plasma |
Table 1. Proteins in transmembrane proteins with NAD (P)-binding Rossmann-fold domains superfamily.
Creative Biostructure has rich professional experience in high quality transmembrane proteins with NAD (P)-binding Rossmann-fold domains production using detergent-free membrane protein expression system, we can perform various strategies for Mempro™ detergent-free protein production, including:
- Mempro™ Transmembrane Proteins with NAD (P)-Binding Rossmann-Fold Domains Production Using Nanodiscs
Nanodiscs are made up of membrane scaffold proteins (MSPs), which are embellished versions of the major ingredient of HDL, apolipoprotein A1 (apoA1). Compared with bicelles, detergent micelles and liposomes, a further native environment where membrane proteins remain monodisperse and active can be offered. Various membrane proteins systems enable to be self-assembled into nanodiscs.
- Mempro™ Transmembrane Proteins with NAD (P)-Binding Rossmann-Fold Domains Production Using Amphipols
Amphipols are a new class of surfactants, they are synthesized by forming amide bonds between octylamine and, somewhile sopropylamine. The wide resources of polymer chemistry can make it possible to tailor the physicochemical properties of amphipols toward peculiar applications in biophysics and biochemistry.
- Mempro™ Transmembrane Proteins with NAD (P)-Binding Rossmann-Fold Domains Production Using Poly (styrene-co-maleic acid) Lipid Particles (SMALPs)
Using SMALPs can make detergent-free isolation of membrane proteins and retention of their crude lipid environment available. Recently, electron microscopy has quickly progressed, which leads to the highly development of data acquisition, together with the conformational and resolution information getable.
These novel detergent-free technologies for transmembrane proteins with NAD (P)-binding Rossmann-fold domains production can be obtained easily, and enabling more comprehensively structural and functional studies.
Creative Biostructure provides other various Mempro™ membrane protein production services. Please feel free to contact us for a detailed quote.
References:
Hille, B. (1978). Ionic channels in excitable membranes. Current problems and biophysical approaches. Biophysical Journal, 22(2), 283.
Jamshad, M., Lin, Y.-P., Knowles, T. J., Parslow, R. A., Harris, C., Wheatley, M., . . . Overduin, M. (2011). Surfactant-free purification of membrane proteins with intact native membrane environment. Biochemical Society Transactions, 39(3), 813-818.
Tribet, C., Audebert, R., & Popot, J.-L. (1996). Amphipols: polymers that keep membrane proteins soluble in aqueous solutions. Proceedings of the National Academy of Sciences, 93(26), 15047-15050.
Bayburt, T. H., Grinkova, Y. V., & Sligar, S. G. (2002). Self-assembly of discoidal phospholipid bilayer nanoparticles with membrane scaffold proteins. Nano Letters, 2(8), 853-856.