Cyclodextrin-Based Liposomes Production
Using dual approach has emerged as a novel plan to tackle the problems of poor aqueous solubility and low rate of dissolution in drug formulation. Therefore, Creative Biostructure combines the cyclodextrins and nanotechnology to develop cyclodextrin-based liposomes that deliver hydrophobic drugs.
How Cyclodextrin-Based Liposomes Work
Cyclodextrins (CDs) are cyclic oligosaccharides formed from glucopyranose units. With hydrophobic cavity inside and hydrophilic outside, they can form complexes with hydrophobic compounds. The CD/drug inclusion complex provide an alternative approach to encapsulate hydrophobic drugs in the aqueous phase of liposome instead of incorporating them into the phospholipids membrane bilayers. This double-loading technique provides better therapeutic effects in the following aspects.
- By complexing with the cyclodextrin and their derivatives, the solubility, stability, and bioavailability of drug can be improved, along with reduced toxicity and modified physicochemical characteristics.
- Cyclodextrin-based liposomes increase the drug loading capacity and entrapment efficiency, prolong the existence of the drug in systemic circulation, and provide controlled, sustained, or targeted release. These liposomes also prevent the dissociation of CD/drug complex and renal excretion of cyclodextrins.
- Relatively low cost.
Cyclodextrin-Based Liposomes Applications
Cyclodextrin-based liposomes is a promising carrier system to preserve volatile and hydrophobic drugs; and provide an access to explore their applications in pharmaceutical, cosmetic and food industry. In addition to liposomes, cyclodextrin-based nanocarriers could be expanded by combination with other carriers like niosomes, nanosponges, micelles, Short Interfering RNA, etc. This complementary carrier system could broaden the scope and accessibility of present and future therapeutic agents.
Creative Biostructure presents our well-developed cyclodextrin-based liposomes technology, which offers the best, customized service to your research:
- Various Cyclodextrins Supply: Cyclodextrins and cyclodextrin derivatives are immediately available off-the-shelf; Customized CDs are synthesized to meet different drug properties.
- Analytical Services: Encapsulation efficiency (EE%), average particle size (PS), polydispersity index (PDI), zeta potential (ZP), liposomal morphology, drug release studies, and in vivo evaluation.
- Cyclodextrin/drug Complexes Preparation Method Development.
- Cyclodextrin/drug Loading in Liposomes Method Development.
- Pharmaceutical Development
- Non-Pharmaceutical Development
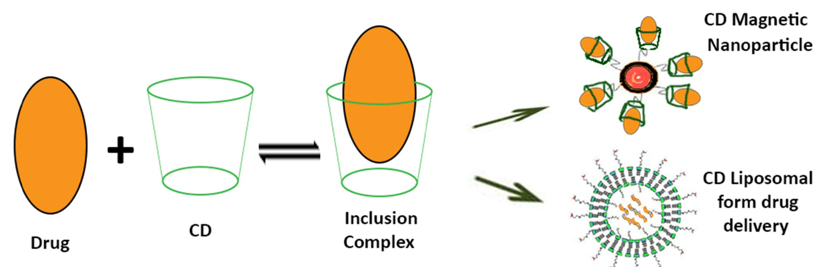 Figure 1. Cyclodextrin-based liposomes (International Journal of Pharmaceutics. 2014)
Figure 1. Cyclodextrin-based liposomes (International Journal of Pharmaceutics. 2014)
Several anti-cancer and anti-inflammatory drugs are developed using cyclodextrin-based liposomes technology. Creative Biostructure offers an advanced liposome manufacturing platform to promote the development of novel drugs. Having an excellent record of designing, producing, and analyzing CD-based liposomes, the quality of our service is endorsed by hundreds of customers.
Please contact us for more details and find out how it helps to the new product development.
Ordering Process
References
- Nadiah Zafar, Hatem Fessi, Abdelhamid Elaissari. (2014) Cyclodextrin containing biodegradable particles: From preparation to drug delivery applications. International Journal of Pharmaceutics. 461: 351-366
- Bina Gidwani, Amber Vyas. (2015) A Comprehensive Review on Cyclodextrin-Based Carriers for Delivery of Chemotherapeutic Cytotoxic Anticancer Drugs. BioMed Research International. doi: http://dx.doi.org/10.1155/2015/198268
- Gharib, R., Auezova, L., Charcosset, C., Greige-Gerges, H., (2016) Drug-in-cyclodextrin-inliposomes as a carrier system for volatile essential oil components: application to anethole, Food Chemistry, http://dx.doi.org/10.1016/j.foodchem.2016.09.110
