Custom MemPro™ Acyl-CoA-binding Protein (ACBP)
Creative Biostructure has developed custom MemPro™ gene-to-structure services for ACBP in the protein expression, purification, structure determination and functional analysis.
ACBP is a highly conserved cytosolic protein with an approximate size of 10kDa, consisting of 86 ~ 103 residues. It has been isolated from a wide range of species including yeasts, plants, reptiles and human, acting as an intracellular acyl-CoA transporter and pool former. In mammals, in addition to the first characterized ACBP in liver (L-ACBP), homologous distinct ACBP-like genes have also been identified in testes (T-ACBP) and brain (B-ACBP). There is yet another group of possibly membrane-bound ACBPs with longer sequences (with up to 533 amino acids).
ACBP is capable of inducing medium-chain acyl-CoA synthesis and binding long-chain acyl-CoA esters with high affinities. However, it shows no affinity towards non-esterified fatty acids or free CoA, revealing that both hydrophilic and hydrophobic interactions are involved in the binding. ACBP is also known as the diazepam binding inhibitor(DBI) orendozepine(EP) because of its ability to displace diazepam from theγ-aminobutyric acid (GABA) receptors. Thus, ACBP is suggested to act as an euro transmitter or neuromodulator, modulating the action of the GABA receptors.
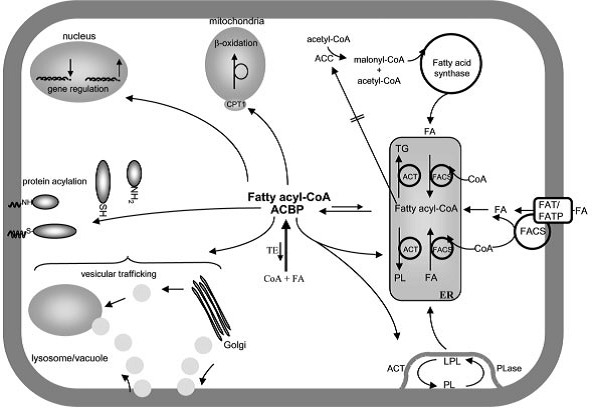
Although ACBP exists as a completely independent protein, intact ACBP-like domains have also been identified in a number of large, multifunctional proteins such as large membrane-associated proteins with N-terminal ACBP domains. ACBP is folded into four α-helix–bundle protein. The binding site locates in a hydrophobic groove on the surface of ACBP, and the ligand can interact with specific residues (conserved positive residues) on the rim of the binding cavity.
Creative Biostructure can provide custom MemPro™ gene-to-structure services for membrane proteins.
