Custom MemPro™ Ammonia and Urea transporters Services
Creative Biostructure provides custom gene-to-structure services for ammonia and urea transporters.
Ammonia and urea transporters (Amt/Mep), alternative name is Rhesus proteins are ubiquitously distributed in gills of fish and rumen epithelium. Human Rhesus (Rh) family proteins can be divided into two functionally distinct groups: the ammonia transporting Rh glycoproteins (RhAG, RhBG, and RhCG) and the nontransporting (nonglycosylated) Rh proteins (RhD and RhCE). Whereas RhCE, RhD and RhAG are erythroid specific, RhBG and RhCG are expressed in key organs like liver and kidney that associated with ammonium transport and metabolism.
The Rh blood group system is one of 35 current human blood group systems and is the second most important blood type, after ABO. RhAG together with Rh proteins, which carry the blood group antigens D and CE, form the core of the Rh complex, which includes glycophorin B, CD47, intercellular adhesion molecule-4 (LW), and AE1. The absence of RhAG will cause Rh-deficiency syndrome that is characterized by abnormalities of the red cell shape and deficiencies of the cation content. The commonly called Rh factors, Rh+ or Rh- refers to RhD antigen. Since D antigen is highly immunogenic, individuals who is Rh- will produce numerous anti-D antibodies when encountered the D antigen on transfused RBCs or fetal RBCs. These response results in the disintegration of RBCs and finally leads to hemolytic anemia. Particularly, a mother who is Rh- may develop antibodies to an Rh+ baby, it will cause severe impairment to the baby’s brain or even death in the fetus or newborn (HDN).
Human RhCGs are found in squamous epithelial tissues such as brain, liver, gut and kidney. They are responsible for pH regulation and ammonia excretion.
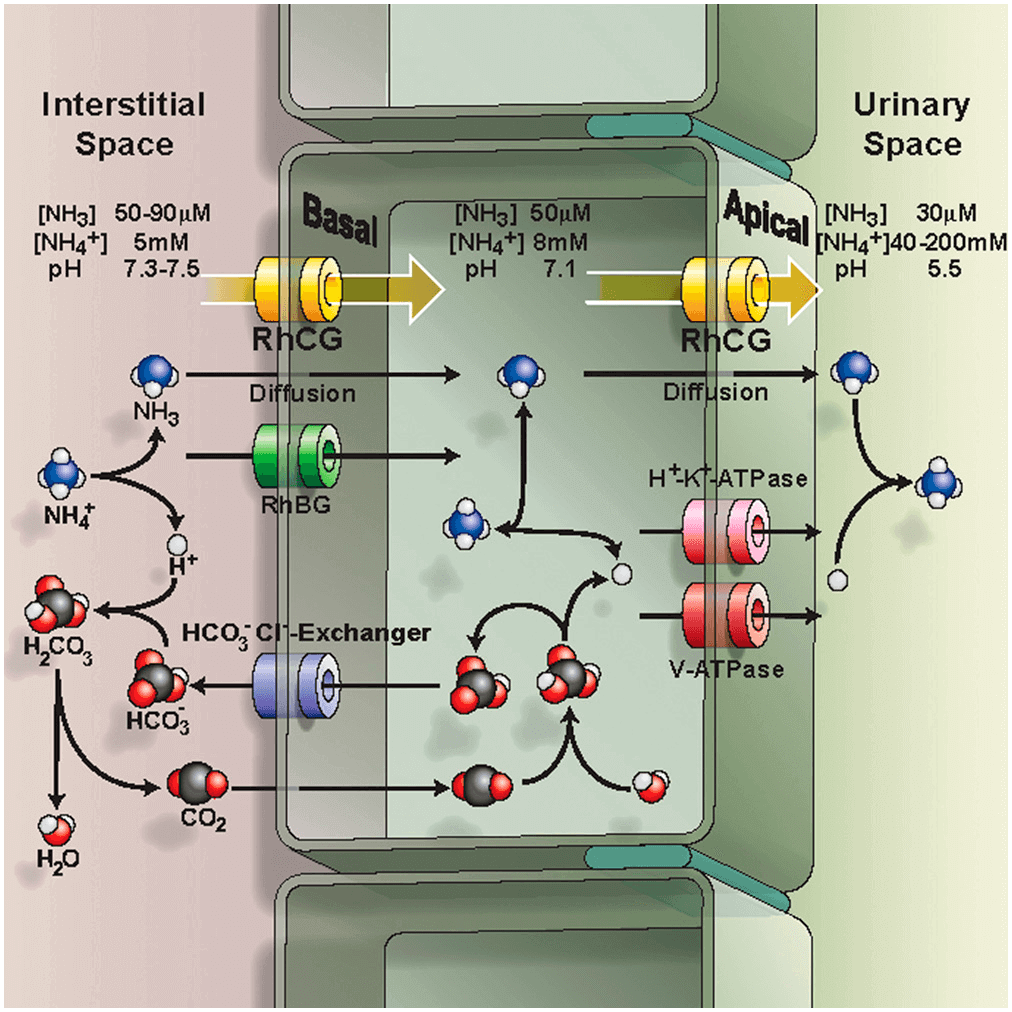 Figure 1. Epithelial model for acid-secreting intercalated cells
Figure 1. Epithelial model for acid-secreting intercalated cells
RhCG and RhBG are extensively expressed on many epithelial cells, and in kidney, they are found in the cells of the collecting tubule and collecting duct acid-secreting intercalated cells as well as in the majority of the cells of the distal collecting tubule. In physiological pH, ammonium ion (NH4+) form predominates while the cell membrane is impervious to charged ionic form. RhCG coupled with H+/K+-ATPase and HCO3-/Cl- exchanger to regulate homeostasis. NH4+ secretion in the kidneys is critical to systemic acid-base homeostasis. RhCG knockout mice show abnormal acidification of the blood since it is unable to remove NH4+ from the urinary. In humans, inability to appropriately dispose of excess H+ in urine results in distal renal tubular acidosis that depresses arterial blood pH (<7.35), and in extreme cases results in kidney stones, rickets, and renal failure. Thus, RhCG plays vital role in renal transport for human physiology.
References:
Gruswitz F, Chaudhary S, Ho J D, et al. Function of human Rh based on structure of RhCG at 2.1 Å[J]. Proceedings of the National Academy of Sciences, 2010, 107(21): 9638-9643.
Zidi-Yahiaoui N, Mouro-Chanteloup I, D'ambrosio A M, et al. Human Rhesus B and Rhesus C glycoproteins: properties of facilitated ammonium transport in recombinant kidney cells[J]. Biochemical Journal, 2005, 391(1): 33-40.
Dean L. Blood groups and red cell antigens[J]. 2005.