Custom MemPro™ B-cell Lymphoma 2 (Bcl-2) Family
Creative Biostructure has developed custom MemPro™ gene-to-structure services for Bcl-2 family. There are about 25 genes encoding proteins of Bcl-2 family up to now, which are highly homologous and contain conserved Bcl-2 homology (BH) domains (e.g., BH1, BH2, BH3 and BH4 domains). The members of Bcl-2 family mainly regulate mitochondrial outer membrane permeabilization (MOMP) and can be functionally categorized functionally into three groups with different structure and function: BH3-only proteins, anti-apoptotic factors (pro-survival cell guardians) and pro-apoptotic factors. The most prominent members of anti-apoptotic factors include Bcl-2,Bcl-XL, Bcl-W, Mcl-1, and CED9. Pro-apoptotic factors mainly include Bax, Bak, Bcl-XS, Bad, Bik and Bid.
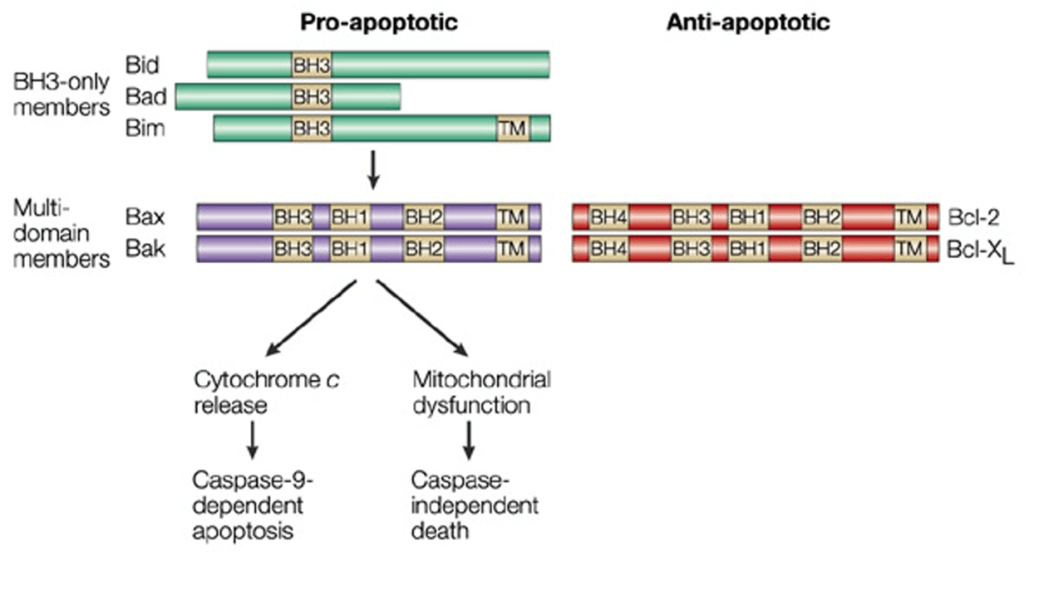 Figure. Bcl-2 family
Figure. Bcl-2 family
Structurally, the anti-apoptotic factors (pro-survival cell guardians) BCL-2, BCL-W, BCL-XL, MCL1 and BCL-B have four BH domains and share similar structures: a central hydrophobic core helix surrounded by a helical bundle, which generate a hydrophobic surface groove. Pro-apoptotoc factors mostly contain BH1, BH2 and BH3 domains while BH3-only proteins are mostly intrinsically disordered. BID is a structural exception of BH3-only proteins which folds similarly with the multi-BH domain members. BH3-only proteins perform pro-apoptotic function by two mechanisms: to activate pro-apoptotic effectors including BAX and BAk; to neutralize anti-apoptotic proteins, which also indicate that different BCL-2 family members have interactions. Neutralization of the anti-apoptotic proteins by BH3-only proteins has been studied well. Their amphipathic helix can bind to the anti-apoptotic proteins directly. And for apoptotic regulation, there is a simple competition, whereby increased BH3-only protein levels, resulting in increased binding of anti-apoptotic proteins, prevent the inhibition of activated BAX or BAK by anti-apoptotic proteins. Additionally, anti-apoptotic proteins neutralize BH3-only proteins to prevent apoptosis, resulting in activating BAX and BAK. BCL-2 family members are diverse functionally in both normal physiology and pathological settings. BCL-2 proteins have many non-apoptotic roles including autophagy, mitochondrial fission and fusion and mitochondrial function. For instance, BCL-2 ablation results in the death of renal epithelial progenitor cells and depleting melanocyte progenitors and mature lymphoid cells, causing fatal polycystic kidney disease. While concomitant loss of BIM, a BH3‑only protein, rescues all of these defects. In consideration the function of anti-apoptotic BCL-2 family members, activation of these factor-regulated pathways can be a candidate strategy for the therapy of many diseases. Additionally, it has shown that BH3 mimetics have great potential in cancer therapy and other BCL-2 members have application in autoimmune and infectious diseases.
Creative Biostructure can provide custom MemPro™ gene-to-structure services for membrane proteins. Please click for more information.
Jesenberger V, Jentsch S. Deadly encounter: ubiquitin meets apoptosis. Nat Rev Mol Cell Biol. 2002 Feb; 3 (2): 112-21.
Peter E. Czabotar, et al. Control of apoptosis by the BCL-2 protein family: implications for physiology and therapy. Nat Rev Mol Cell Biol. 2014 Jan;15(1):49-63.