HA assay for Mempro™ Virus-like Particles (VLPs) Characterization
Based on our Mempro™ Membrane Protein platform, Creative Biostructure has developed an advanced and unparalleled virus-like particles (VLPs) platform in recent years. VLPs are non-infectious virus particles which can be self-assembled, providing a great potentials as novel vaccine candidates against various viral infections. Some VLP-based human influenza vaccines have been already proved to be effective because of the high immunogenicity and safety. The hemagglutination (HA) assay is one of our major techniques to qualify the virus-like particles.
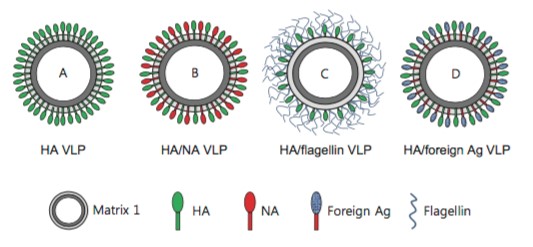 Figure 1. Schematic diagrams of influenza virus-like particles (VLPs). (Biotechnology and Bioengineering, 2014)
Figure 1. Schematic diagrams of influenza virus-like particles (VLPs). (Biotechnology and Bioengineering, 2014)
Characterization of VLPs plays an essential role in VLP-based vaccine development. Poor characterization of VLPs candidates affects both VLP design and bioprocess development, and leads to misinterpretation of the immunological data. Creative Biostructure has focused on the better VLPs characterization of their functionalities and the physical properties including the size and polydispersity.
The HA assay was firstly to characterize the influenza virus-like particles based on the agglutination property. Creative Biostructure has modified and applied this method for the HA activity determination in VLPs preparations. The basis of agglutination is the sialic acid binding ability of HA on erythrocytes (RBC). This is crucial for influenza virus because the virus must bind to sialic acid to enter the cell and begin the infection cycle. Although this point is not significant for VLPs since they are non-infectious, their ability to bind sialic acid shows that HA is in the correct conformation and active.
The HA assay measurement is based on the observation of the agglutination of RBCs by the HA protein. The principle of HA assay is very simple, but the preparation is laborious and has some drawbacks. Creative Biostructure has established the quantification of virus-like particles with electron microscopy based on the RBC concentration that there is approximately one influenza virus for each red blood cell at the end point of agglutination.
Besides HA assay, Creative Biostructure can also offer a broad range of Mempro™ Virus-like Particles (VLPs) Characterization services, such as AFFFF-MALS (asymmetric flow field-flow fractionation with multi-angle light scattering detection), Electrospray differential mobility analysis (ES-DMA), ELISA, Western blot, RP-HPLC, Electron microscopy (TEM), LC-MS, Dynamic light scattering (DLS), Enzymatic NA activity assay, Surface Plasmon Resonance Immunoassay (SPR), etc. Please feel free to contact us for a detailed quote.
References:
D. Lee. et al. (2014). Progress and hurdles in the development of influenza virus-like particle vaccines for veterinary use. Biotechnology and Bioengineering. 3:133–139.
C. M. Thompson, et al. (2013). Analytical technologies for influenza virus-like particle candidate vaccines: challenges and emerging approaches. Virology Journal,10: 141.
X. Tang, et al. (2010). Hemagglutinin displayed baculovirus protects against highly pathogenic influenza. Vaccine, 28: 6821–6831.