Custom MemPro™ Ion Channel Services
With advanced techniques and rich experience in structural biology field, Creative Biostructure now provides custom MemPro™ Ion channels structure-function services for structure-based functional studies of ion channels. Our professional technical team can serve you from gene to structure, especially in construction, purification, crystallization, data collection, structure determination and functional analysis.
Ion channels are important transmembrane (TM) pore-forming proteins activated by a variety of stimuli including ligand binding, voltage change and stretch. When channels are activated, selected ion flows across the membrane result in voltage changes that are crucial for many physiological processes (e.g., nerve conduction, epithelial secretion, muscle contraction and sensory reception). The hereditary gene mutations of ion channels can lead to channelopathies such as epilepsy, cardiac arrhythmia, migraine, asthma, hyperekplexia, blindness, deafness, diabetes, hypertension, irritable bowel syndrome and cancer. Moreover, non-genomic ion channel modulation is associated with a broad range of diseases including cardiac disorders, neurological indications, neuropathic, renal disorders and inflammatory pain. All these have made ion channels the major therapeutic targets.
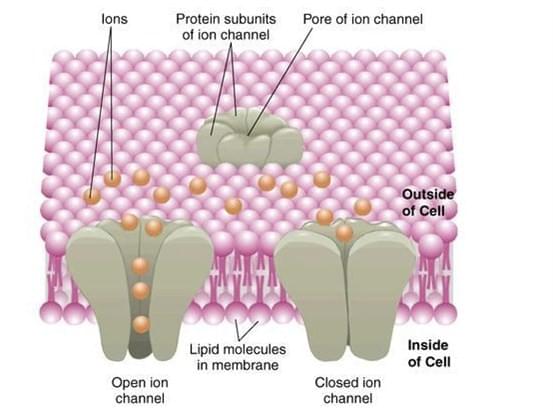
Ion channel
Detailed structural studies have shown that ion channels are in a constant thermal motion status and display conformational heterogeneity all the time. Activation stimuli, including voltage changes or ligand bindings, initiate a conformational wave that propagates throughout the protein to the gate. And single channel recording can resolve movements at the gate with a high temporal resolution. Site-directed mutagenesis or protein modifications can provide important insights into their structural and functional complexities. However, the conformational dynamics underlying opening, closing, desensitization and drug modulatory mechanisms are still unknown due to the difficulty of real-time conformational change monitoring in domains farther from the gate.
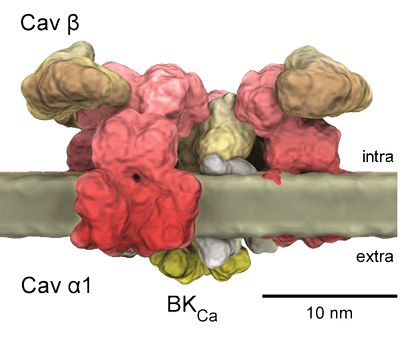
Structural diagram of Cav channels.
The α-interacting domain (AID; shown in red on the I–II linker) binds to the Cavβ subunit with high affinity. The pore-forming loops between TM segments S5 and S6 are marked with “P”, and the voltage-sensor domain in TM segment S4 is indicated by “+” signs.
Structural biology techniques including X-ray crystallography, Cryo-Electron Microscopy (Cryo-EM) and Nuclear Magnetic Resonance (NMR) spectroscopy have determined the structures of some channels. But it is usually difficult to assign structural configurations to the conformational states, if only inferred from the electrophysiological analysis. Besides, the structures of some important ion channels are still unknown.
Ion channels play important roles in drug discovery and therapy. Techniques like X-Ray, NMR and Cryo-EM are used for structure determination; meanwhile, computer modeling can be used to study the structure-function relationship of ion channels across the superfamily. Creative Biostructure can serve you effectively from gene to structure with functional analysis for different ion channels.
Guarantees:
- Quick assessment and optimization of targets for construction, high level expression, stability and crystallizability.
- Panel of purified protein assays to facilitate crystallization, crystal optimization and structure determination.
- Customized screening methods.
- High QA/QC standards.
- Data reporting: receive complete standard or customized reports for seamless integration into in-house databases.
- Functional screening: follow ups on hits in any GPCR function assays.
Learn more information about our MemPro™ membrane protein services.