Custom MemPro™ Services for Amyloid beta (A4) Protein
Creative Biostructure has developed custom MemPro™ gene-to-structure services for Amyloid beta (A4) in the protein expression, purification, structure determination and functional analysis.
Amyloid beta (Aβ or Abeta), consisting of 36 ~ 43 amino acids, is suggested to be crucial in Alzheimer's disease (AD), which is the most common form of dementia in elderly. The cause of AD is unknown. However, the ubiquity and cytoxicity of aggregated Aβ peptide in Alzheimer’s patients have indicated a link between the misfolding of Aβ peptide and the ultimate upstream cause of AD.
Though many studies demonstrate the absence of Aβ does not lead to any loss of physiological functions, Aβ has been found to be associated with several biochemical processes, including activation of kinase enzymes, protection against oxidative stress, regulation of cholesterol transport, functioning as a transcription factor and anti-microbial activity. For instance, 1) extracellular Aβ can be transported into cell under oxidative and heat stress; 2) Aβ can induce an increase in the expression levels of apoptosis–associated tumor protein 53 and transcription factor ASCL1; 3) Aβ interacts directly with specific DNA sequences in the regulatory regions of AD-associated genes. Therefore, Aβ is believed to play a central role in the development of AD pathology, although the cause of AD is much complicated and needs more investigations.
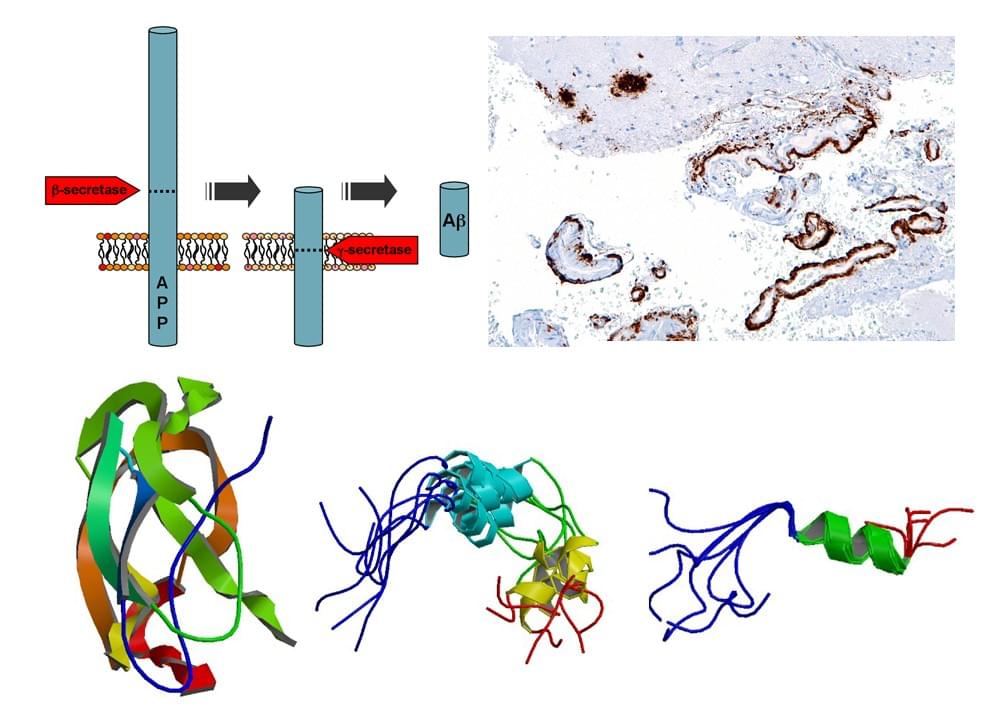
A distinctive feature of AD is the accumulation of amyloid plaque in the affected brains with Aβ peptide as the major constituent of the plaque. Aβ peptide is derived from the cleaving of its much larger amyloid precursor protein (APP) by β- and γ-secretase in the amyloidogenic pathway. As Aβ is neurotoxic in monomers and oligomers, several intervention strategies either to block Aβ formation or recognize and attack formed Aβ have been developed, such as using β-Secretase inhibitors or/and γ-Secretase inhibitors, immunotherapy, chelation therapy and drug therapy.
Creative Biostructure can provide custom MemPro™ gene-to-structure services for membrane proteins.