Custom MemPro™ TPR Alpha-Helical Repeat Proteins
Creative Biostructure has developed custom MemPro™ gene-to-structure services for TPR alpha-helical repeat proteins in the protein expression, purification, structure determination and functional analysis.
The tetratricopeptide repeat (TPR), consisting of 3-16 tandem repeats of 34 amino acid residues, is a structural motif found in a wide range of proteins. The TPR was identified and named in 1990, and from then on, many structures of TPR domains have been solved. TPR domains serve as interaction modules and multiprotein complex mediators; hence, they are crucial for many biological functions.
To date, more than 5,000 TPR-containing proteins have been identified in different organisms, and at least 100 structures have been solved and deposited in the Protein Data Bank (PDB), revealing at a molecular level the versatility of these motifs. The first solved TPR structure is the protein phosphatase 5 (PP5), and its TPR is composed of a pair of antiparallel alpha helices. TPR adopts a helix-turn-helix arrangement with adjacent TPR motifs packed in a parallel fashion, resulting in a spiral of repeating anti-parallel alpha-helices. The packing angle between two helices, helix A and helix B, is ~24°within a single TPR and generates a right-handed superhelical shape.
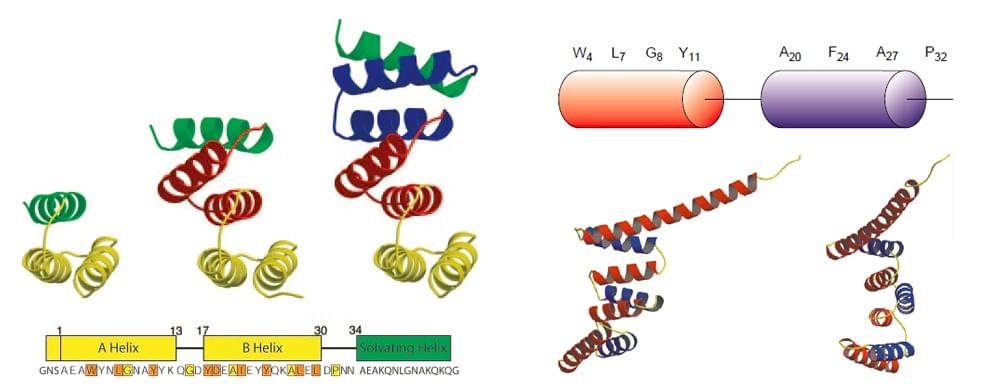
The basic function of TPR domains is to mediate protein–protein interactions, and this can be achieved in a variety of ways. The elegant superhelical fold of TPR-containing proteins presents several binding surfaces for the formation of multiprotein complexes. Given that the basic TPR scaffold can be redesigned to modulate the binding specificity and/or affinity toward target peptide ligands, TPR-containing proteins represent a bright prospect for protein engineering, therapeutics, and biotechnology. Moreover, TPR-containing proteins can also bind effectively with designed ligands of high affinities, serving as scaffolds for protein presentation in nano technological applications.

Creative Biostructure can provide custom MemPro™ gene-to-structure services for membrane proteins.