MemPro™ Rhodopsin-like Receptors Services
Rhodopsin-like receptors are G-protein coupled receptors (GPCRs) which is the largest class of membrane protein in human genome. Based on their unique seven-transmembrane segments, scientists predict there are approximately 800 GPCRs in human while most structures and functions remained unknown. It widely expressed in many kinds of cell surfaces for signaling of diverse group of ligands to heterotrimeric G proteins. Ligands range from subatomic particles, to ions, to small organic molecules, to peptides and proteins binding to different domains cause a conformation change that trigger signaling via guanine nucleotide-binding proteins (G proteins) and modulates the activity of down-stream effectors. Though they share no significant sequence similarity with each other and regulate a wide range of functions, they have conserved 7-TM structure and transduce extracellular signals through interactions with G proteins.
Rhodopsin-like GPCR, also known as class A GPCR, is the largest class of GPCR, which account for approximately 85% of GPCR genes. The other GPCRs include secretin-like GPCR, the metabotropic glutamate GPCR, fungal mating pheromone receptors, cAMP receptors, and frizzled/smoothened GPCR.
Additionally, Rhodopsin-like GPCRs includes receptors for a wide variety of small molecules, neurotransmitters, peptides and hormones, together with olfactory receptors, visual pigments, taste type 2 receptors and five pheromone receptors. There are 19 subgroups in total and most of them have received extensive attentions. They mainly include:
- Chemokine receptors
- Angiotensin II receptors
- Opioid receptors
- Leukotriene receptors
- Orexin receptors
- Vasopressin receptors
- Glycoprotein hormone receptors
- Purinergic receptors
- Lysophosphatidic acid receptors
- Sphingosine 1-phosphate receptors
- Melanocortin/ACTH receptors
- Rhodopsin receptors
- 5-HT receptors
- Adrenergic receptors
- Dopamine receptors
- Histamine receptors.
- Adenosine receptors
- Muscarinic acetylcholine receptors
- Olfactory receptors
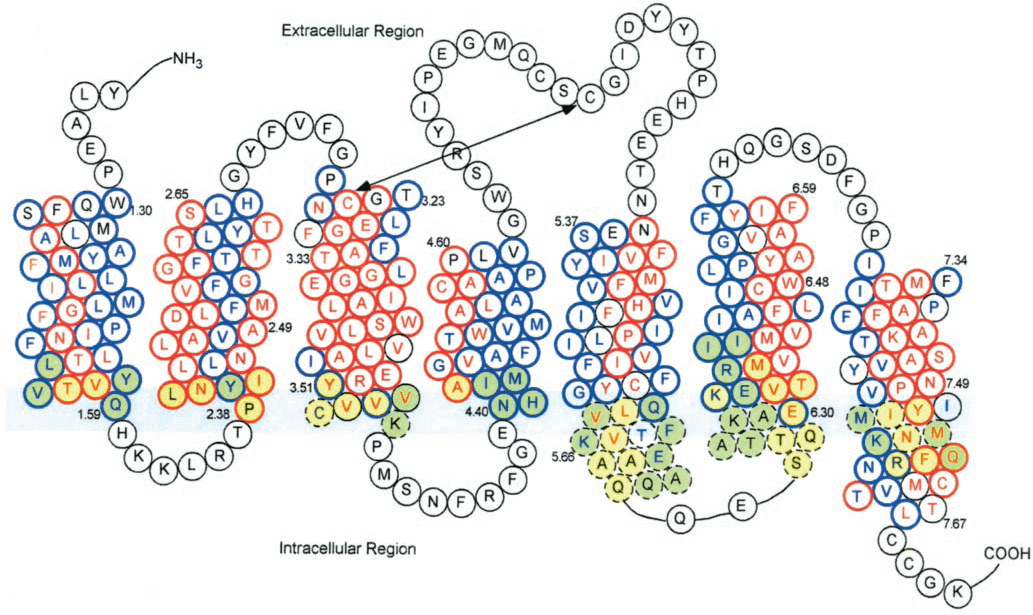 Figure 1. Dopamine receptor——seven transmembrane helix structure.
Figure 1. Dopamine receptor——seven transmembrane helix structure.
Since rhodopsin-like receptors play essential role in a variety of biological process through a single target, a lot of researches have been approached to develop new therapeutic strategies. It has been estimated that about 50% of all modern drugs act on GPCRs. Recently, many X-ray structure have shown a direct involvement of water molecules in ligand binding GPCR, that influencing the ability of β-arrestin signaling. Thus, one GPCR can signal through multiple different downstream pathways beyond traditional G-proteins. They can respond to different ligands and active different signaling pathway to different extents.
GPCRs have 7 transmembrane regions which makes structure elucidation much more difficult. Creative Biostructure has years of experience in membrane protein expression in established systems and we also utilized X-ray crystallography, NMR spectroscopy and cryo-electron microscopy for structure determination. These services enable you to
- Harvest purified, functional membrane proteins
- Get insight of TM segments and predict protein functions.
- Discover new drug targets and ligands design.
References:
Birdsall N J M. Class A GPCR heterodimers: evidence from binding studies[J]. Trends in pharmacological sciences, 2010, 31(11): 499-508.
Tautermann C S. GPCR structures in drug design, emerging opportunities with new structures[J]. Bioorganic & medicinal chemistry letters, 2014, 24(17): 4073-4079.